

Journal of Pharmaceutical Research
DOI: 10.18579/jopcr/v22.1.23.7
Year: 2023, Volume: 22, Issue: 1, Pages: 50-54
Original Article
Kamal Nayan Dwivedi 1 , Anurag Mishra 1 , Rajesh Kumar Singh ✉ 1
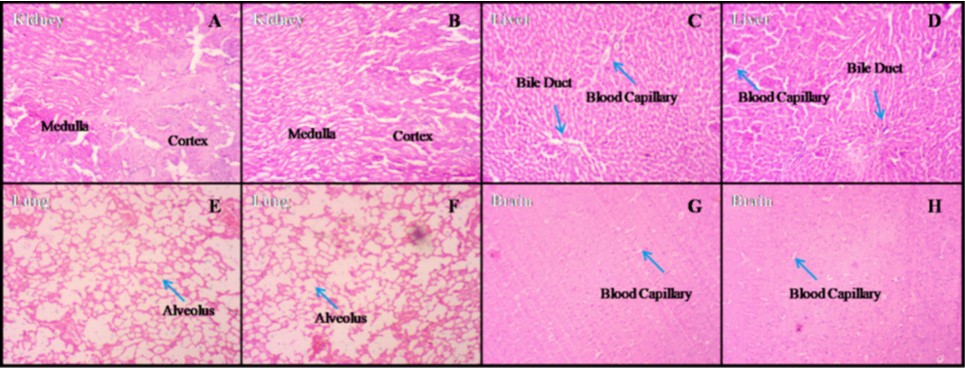
A polyherbal formulation consisting of Withania somnifera, Tinospora cordifolia, Moringa oleifera, Adhatoda vasica, Pipper longum, Glycyrrhiza glabra, Ocimum sanctum and Curcuma longa has been used for Covid-19 treatment empirically, which was found effective. However, toxicity data were not available for this polyherbal formulation. This study aims to assess the polyherbal formulation's oral acute and subchronic toxicity in rats. The fixed-dose approach was used to conduct the acute toxicity investigation on 6 female Wistar rats for the treatment group and 5 female Wistar rats for the control group. A single dosage of this polyherbal formulation weighing 2,000 mg/kg was administered orally to the test group. At the end of the investigation, no fatalities or major toxic effects were noted, and it was determined that the lethal dose 50% (LD50) of the polyherbal formulation was greater than 2,000 mg/kg. Vital organs underwent macroscopic and microscopic inspection, neither of which revealed any toxicity signs. The polyherbal formulation was given orally for 91 days during the subchronic toxicity research in dose variations: 250 mg/kg, 500 mg/kg, 1,000 mg/kg, 2,000 mg/kg, and 4,000 mg/kg. The daily dose for a human is the same at the lowest level of 250 mg/kg. On physical signs and symptoms, weight growth, food intake, haematological parameters, biochemical parameters, and macroscopic and microscopic examination of organs, no major harmful effects were seen at any of these doses. These results demonstrated that the oral administration of this polyherbal formulation over the short- and long-term is safe when taken as suggested.
Keywords: Ayurvedic, Polyherbal, COVID19, Acute toxicity, Subchronic Oral Toxicity
© 2023 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.