

Journal of Pharmaceutical Research
DOI: 10.18579/jopcr/v21i4.22.15
Year: 2022, Volume: 21, Issue: 4, Pages: 144-150
Original Article
A Suneetha ✉ 1 , M Vijaya Lakshmi 1 , K Jyothi 1 , B Yoga Priyanka 1
Corresponding author ✉: [email protected]
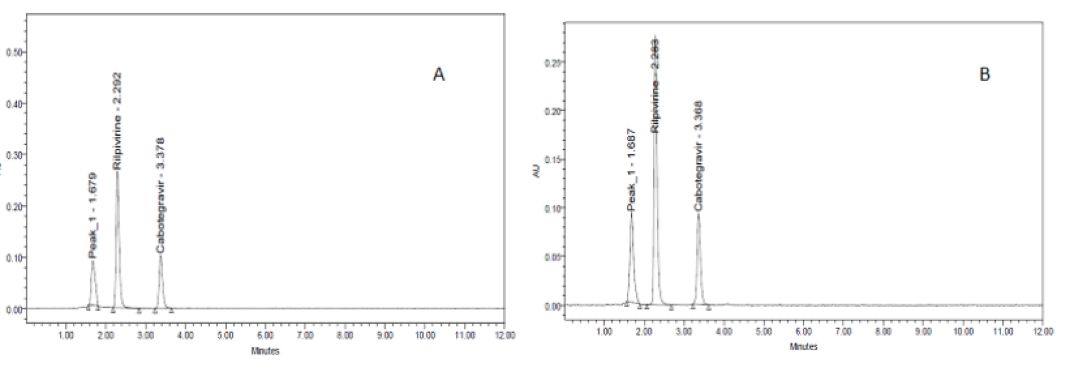
A simple, sensitive, specific, accurate, and stability-indicating reversed phase high performance liquid chromatographic method was developed for the simultaneous determination of cabotegravir and rilpivirine in injection dosage form, using a Waters Model No.2695 series compact system fitted with Agilent - C18 column (BDS) (150 X 4.6 mm, 5μm) and a mobile phase composed of 0.01N KH2PO4 buffer (pH: 4.8): acetonitrile (70:30v/v). The 260 nm wavelength was chosen. The retention times of cabotegravir and rilpivirine were found to be 2.30 and 3.187 minutes, respectively. Linearity was established for cabotegravir and rilpivirine in the range of 25-150 µg/mL and 37.5-225µg/mL respectively: with correlation coefficient (r2) of 0.9999 for both the drugs. Cabotegravir and rilpivirine were found to have %RSDs of 0.4 and 0.3 respectively, for system precision. The proposed methods intra- and inter-day precision assessments showed a relative standard deviation (%RSD) below the maximum permitted level of 2.0. Accuracy was carried out in triplicate, and the percentage recovery was 100.25% for cabotegravir and 99.79% for rilpivirine, respectively. The limit of detection and limit of quantification for cabotegravir were found to be 0.24 and 0.74 μg/mL, respectively and 1.10 and 3.34 μg/mL, for rilpivirine respectively. Both medications were put to a range of stress conditions, including thermal, acidic, basic, oxidative, and photolytic stress. The findings demonstrated that, with the exception of heat, UV, and neutral environments, considerable degradation was found in acidic, basic, and oxidative conditions where a good separation of drug peaks was seen in the presence of the degradation products. As a result, this technique can be used to quantitatively to analyze cabotegravir and rilpivirine in bulk drug and injection dosage forms.
Keywords: Cabotegravir, RP HPLC, Rilpivirine, ICH guidelines
© 2022 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.