

Journal of Pharmaceutical Research
DOI: 10.18579/jopcr/v25.i1.173
Year: 2026, Volume: 25, Issue: 1, Pages: 8-15
Review Article
Shivali Rahi1*, Jayamanti Pandit2
1Department of Regulatory Affairs, School of Pharmaceutical Sciences, MVN University, Palwal – 121105, Haryana, India
2Department of Pharmaceutics, School of Pharmaceutical Sciences, MVN University, Palwal – 121105, Haryana, India
*Corresponding Author
Shivali Rahi
Email: [email protected]
Phytopharmaceuticals bridge traditional herbal medicines and modern pharmaceuticals by combining plant-based bioactives with scientific validation. India, endowed with rich biodiversity and traditional medicine heritage, holds immense potential in this field. However, regulatory ambiguity, lack of standardization, and weak global alignment continue to hinder growth. The objective of the study is to evaluate India’s current regulatory framework for phytopharmaceuticals, identify key challenges, and assess recent initiatives aimed at fostering innovation and harmonization with international standards. A qualitative analysis of regulatory documents, government notifications, and policy guidelines was conducted. Comparative evaluation was performed between India’s Drugs and Cosmetics Act and Rules with international frameworks such as WHO, ICH, and FDA to assess regulatory convergence and gaps. India’s regulatory progress includes the 2015 amendment to the Drugs and Cosmetics Rules defining phytopharmaceuticals and the New Drugs and Clinical Trials Rules, 2019, which streamline approvals. Initiatives like the Phytopharmaceuticals Mission, interagency collaborations (ICMR–DBT–CSIR), and inclusion of monographs in the Indian Pharmacopoeia highlight institutional support. Yet, high development costs, overlapping jurisdiction with AYUSH, and undefined preclinical–clinical criteria persist as barriers to market growth. India’s evolving phytopharmaceutical regulations mark a vital step toward integrating traditional wisdom with modern science. Continued policy clarity, quality standards, and international harmonization can establish India as a global leader in evidence-based, plant-derived therapeutics.
Keywords: Phytopharmaceuticals, CDSCO, Regulatory framework, AYUSH, Indian Pharmacopoeia, Herbal medicines, Drug approval, India
Medicinal plants have long served as the cornerstone of traditional healthcare systems around the world, particularly in India, where Ayurveda, Siddha, and Unani systems are widely practiced[1]. The use of traditional herbs for therapeutic purposes has grown in popularity in recent years, opening up prospects for international promotion of Indian traditional medicines[2]. However, rising global demand for scientifically validated plant-derived treatments has resulted in the formation of a new regulatory category in India known as phytopharmaceuticals [3]. Phytopharmaceuticals, as opposed to traditional herbal medications, are standardized plant-derived extracts or fractions containing identified bioactive constituents, with preclinical and clinical data supporting their safety and efficacy[4-6].
The formal recognition of phytopharmaceuticals under the Drugs and Cosmetics Rules through Gazette Notification G.S.R. 918(E) in 2015 marked a significant regulatory milestone. By placing phytopharmaceuticals under the purview of the Central Drugs Standard Control Organization (CDSCO), India created a distinct regulatory pathway separate from AYUSH-regulated traditional medicines. This classification requires phytopharmaceuticals to undergo scientific evaluation comparable to new chemical entities (NCEs), including data on quality, safety, and clinical efficacy[7].
The regulatory environment associated with phytopharmaceuticals is complex in nature, as it aspires to create an equation between traditional knowledge and present pharmaceutical standards. The market for herbal medicine is largely unregulated and has inadequate quality control, thus raising safety issues[8]. The lack of clear policies affects most of the matters developed in different regulatory regimes, including the determination and assessment of safety and effectiveness, quality control, and monitoring of safety, in addition to searching and recording information on alternative medicine and traditional medicine[9].
Challenges such as overlapping regulations with AYUSH, lack of detailed evaluation guidelines, high development costs, and limited harmonization with global botanical drug standards are some of the problems that have made it hard to grow. Therefore, a focused examination of India’s regulatory framework is essential to understand the prospects, barriers, and policy directions required to unlock the country's vast botanical potential[10]. Several well-known phytopharmaceutical products have gained international recognition for their therapeutic benefits. These products are derived from plant-based compounds and are used in various healthcare applications ([Table. 1]).
| Marketed Product | Uses | Brand Name | References |
|---|---|---|---|
| Silymarin (Milk Thistle) | Primarily used for liver diseases, including cirrhosis, hepatitis, and liver damage from alcohol and toxins. | Legalon, Silibinin, Siliphos. | [11] |
| Curcumin (Turmeric) | Known for its anti-inflammatory, antioxidant, and potential anti-cancer properties. | Meriva, Curcumin C3 Complex. | [12] |
| Artemisinin (Sweet Wormwood) | The primary active component in the treatment of malaria. | Coartem (artemether and lumefantrine combination), Riamet. | [13] |
| Vinpocetine | Used for improving cognitive functions and treating cerebrovascular disorders like stroke and memory issues. | Cavinton, Vinpo-10 | [14] |
| Cannabidiol (CBD) | Widely marketed for treating epilepsy (e.g., Dravet syndrome), pain, anxiety, and other conditions. | Epidiolex (FDA-approved), Charlotte’s Web. | [15] |
| Andrographis paniculata (Kalmegh) | Used in respiratory infections, liver issues, and immune system support. | KalmCold,Kan Jang. | [16] |
| Ginkgo biloba | Mainly marketed for cognitive improvement, memory enhancement, and circulatory disorders. | Tebonin,Ginkgold. | [17] |
| Quinine (Derived from Cinchona Bark) | Traditionally used for treating malaria. | Quinate, Qualaquin. | [18] |
| Resveratrol | Promoted for cardiovascular health, anti-aging, and antioxidant benefits. | Longevinex. | [19] |
| Ashwagandha (Withania somnifera) | Adaptogen used for stress relief, cognitive function, and improving vitality. | Sensoril, KSM-66. | [20] |
| Picroliv (Picrorhiza kurroa | Used for liver protection and boosting immune responses. | Liv-52, Picroliv | [21] |
Although several plant-derived products such as artemisinin and cannabidiol have achieved regulatory approval in international jurisdictions (e.g., FDA botanical drug pathway), very few products in India have successfully navigated the CDSCO phytopharmaceutical approval pathway. Numerous herbal products marketed in India remain classified under the AYUSH framework instead of the phytopharmaceutical category. This regulatory disparity underscores the constrained shift from conventional botanicals to evidence-based phytopharmaceuticals within the Indian framework[22].
This article examines the evolution, framework, and implementation of India's phytopharmaceutical regulatory environment, concentrating on legislative provisions,
approval processes, institutional mechanisms, and novel policy initiatives. It assesses how alignment with international botanical medication standards, improved regulatory clarity, and greater institutional coordination might unleash India's tremendous biodiversity potential. From a regulatory point of view, this study shows that India has created a fundamental foundation for evidence-based plant-derived treatments. However, deliberate reforms and strategic alignment are needed to make phytopharmaceuticals a globally competitive pharmaceutical business.
The formal regulatory recognition of phytopharmaceuticals in India represents a significant policy shift toward integrating traditional plant knowledge with modern drug regulation. This amendment defined a phytopharmaceutical drug as a purified and standardized fraction containing at least four bioactive or phytochemical markers derived from a medicinal plant, intended for internal or external use for diagnosis, treatment, mitigation, or prevention of disease[23, 24].
Not like conventional herbal remedies overseen by the Ministry of AYUSH, phytopharmaceuticals are monitored by the Central Drugs Standard Control Organization (CDSCO). By placing phytopharmaceuticals within the framework of the Drugs and Cosmetics Act, 1940, India established a distinct regulatory pathway requiring scientific validation comparable to that of synthetic new drugs. This statutory recognition marked the transition of select botanicals from traditional medicine to evidence-based pharmaceutical products[8, 25].
A key feature of India’s regulatory landscape is the bifurcation between AYUSH-regulated traditional medicines and CDSCO-regulated phytopharmaceuticals. Classical Ayurvedic, Siddha, and Unani formulations are regulated under separate provisions and are not subject to the same clinical trial requirements as phytopharmaceutical drugs[26, 27].
In contrast, phytopharmaceuticals are treated as “new drugs” under Rule 122E of the Drugs and Cosmetics Rules. As a result, manufacturers must submit detailed scientific evidence on quality, safety, and efficacy. This jurisdictional distinction is critical but has also created ambiguity in industry adoption, as products with similar botanical origins may follow different regulatory pathways depending on their development approach and claims.
The regulatory approval process for phytopharmaceuticals aligns closely with the framework applicable to new chemical entities (NCEs). Appendix IB of Schedule Y specifies the data requirements, which include:[28]
Botanical identification and authentication
Standardization of extract and quantification of bioactive markers
Preclinical safety and pharmacology studies
Phase I–III clinical trials to establish safety and efficacy
Submission of a comprehensive dossier for CDSCO review
The New Drugs and Clinical Trials Rules (NDCTR), 2019 further strengthened this pathway by introducing defined timelines for regulatory review and a risk-based approach to clinical trial monitoring. These rules aim to enhance transparency and expedite approval processes while maintaining patient safety standards[29].
Due to the fundamental variation of substances derived from plants, quality control plays a crucial role in phytopharmaceutical regulation. The Indian Pharmacopoeia Commission (IPC) has commenced the incorporation of phytopharmaceutical monographs to set criteria for purity, potency, stability, and marker-based standardization[30].
Compliance with Good Manufacturing Practices (GMP) is mandatory, ensuring uniformity in sourcing, extraction, formulation, and distribution. Quality assurance techniques are vital for attaining regulatory credibility, especially in international trade and worldwide market acceptability[31, 32].
India’s regulatory framework is supported by collaborative institutional initiatives involving the Department of Biotechnology (DBT), the Indian Council of Medical Research (ICMR), and the Council of Scientific and Industrial Research (CSIR). The Phytopharmaceuticals Mission was launched to promote translational research, facilitate industry–academia partnerships, and strengthen infrastructure for plant-based drug development[33-35].
Additionally, efforts are underway to align Indian phytopharmaceutical standards with international frameworks such as the World Health Organization (WHO) guidelines[36] and International Council for Harmonisation (ICH) principles[37]. Such harmonization is crucial for improving export competitiveness and ensuring global regulatory acceptance.
Despite the progress that has been made in regulatory recognition, there are still certain obstacles that need to be conquered. Due to a number of factors, the CDSCO has been unable to receive a large number of applications. These factors include the absence of specific review rules that are specific to phytopharmaceuticals, the perception that these products are comparable to those of AYUSH medicines, the high costs of development, and the limited instances of regulatory oversight.
The legal recognition of phytopharmaceuticals in India is organized, which creates a firm foundation for the nation's future progress. Despite this, the country is still in the process of expanding. A number of variables, including enhanced procedural clarity, stronger harmonization with international botanical drug frameworks, and increased regulatory guidance that is adapted to plant-based complexity, have contributed to India's strategic positioning to expand its evidence-based phytopharmaceutical business. India is in a position to strategically build its phyto- pharmaceutical sector[38].
In the last two decades, demand has surged for phytopharmaceuticals in the international market. It is still challenging to ensure their effectiveness, safety, and quality. Quality assurance must be incorporated at every stage of the development of phytopharmaceutical medicine. Additionally, there is a need to raise awareness among health professionals about the importance of quality, evidence-based research[4]. Despite formal regulatory recognition and institutional support, several structural and operational challenges continue to constrain the growth of phytopharmaceuticals in India.
Uncertainty in Regulations: The regulations have not yet provided a clearly accurate level of toxicity, clinical assessment, or the nature of preclinical and clinical information to be admissible for the consideration of phytopharmaceuticals. For example, such acceptable clinical trial endpoints or toxicological levels have not yet been determined in comparison with synthetics.
High Development Costs and Time: This process is time-consuming and costly because they have to go through the same strict scrutiny as NCEs for regulations. It is a very strict criterion that proves difficult, especially for a small manufacturer, due to the dynamic nature of this industry.
Overlap with AYUSH: Ayurvedic, Siddha, and Unani medications are governed by the Indian Ministry of AYUSH, but the former is not regulated by the same standards of clinical trials that the latter undergoes in control of phytopharmaceuticals. Thus, though both groups make use of plant-based preparations, they are regulated differently, and this has been misinterpreted and overlapping at several instances. Traditional medicines find wide acceptance in India, and these do not share similar clinical requirements, which further challenges the commercial acceptance of phytopharmaceuticals[4].
Standardization: Variability in the composition of plant-based medicines, influenced by factors like geographical location, season, and cultivation practices, leads to inconsistent therapeutic outcomes. This complicates product approval, as regulatory bodies require strict adherence to quality standards, efficacy, and safety, comparable to synthetic drugs[40, 41].
Complex regulatory frameworks: India’s regulations for phytopharmaceuticals, though evolving, remain less streamlined compared to Western countries. The regulatory landscape needs clearer guidelines to support product development and commercialization while ensuring quality and safety. Many phytopharmaceuticals are based on traditional knowledge, making patenting difficult under current legal frameworks. This restricts the development of proprietary formulations and investments in research[42].
Environmental Concerns: As the demand for medicinal plants increases, there's a risk of overexploitation of wild plant populations. Sustainable cultivation and harvesting practices need to be developed and implemented to protect biodiversity.
As of October 2024, India's phytopharmaceutical regulatory system exhibits modest progress but remains in an early consolidation phase. Although the 2015 amendment to the Drugs and Cosmetics Rules that created a distinct category for phytopharmaceuticals, the number of applications submitted to the Central Drugs Standard Control Organization (CDSCO) has remained limited. The restricted acceptance probably reflects the early development stage of many candidates, rigorous evidence criteria similar to those for synthetic drugs, and a growing comprehension of regulatory requirements[43].
The major Indian institutions involved in phytopharmaceutical research are given in [Table. 2].
| Institution | Focus Area | Notable Achievements |
|---|---|---|
| CSIR | Drug discovery from plants | Anti-diabetic drug from Pterocarpus marsupium |
| ICMR | Clinical trials of herbal formulations | Standardization of Ayurvedic formulations |
| IIT Bombay | Phytochemical analysis | Novel extraction techniques for bioactive compounds |
| AIIMS | Clinical studies on herbal drugs | Efficacy studies on traditional remedies |
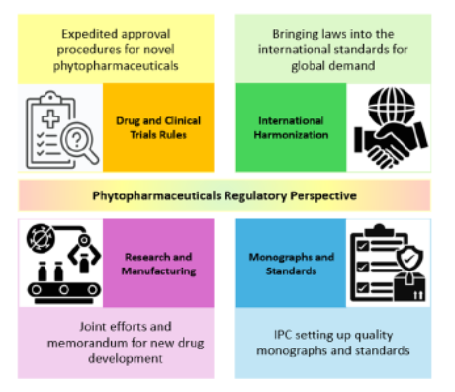
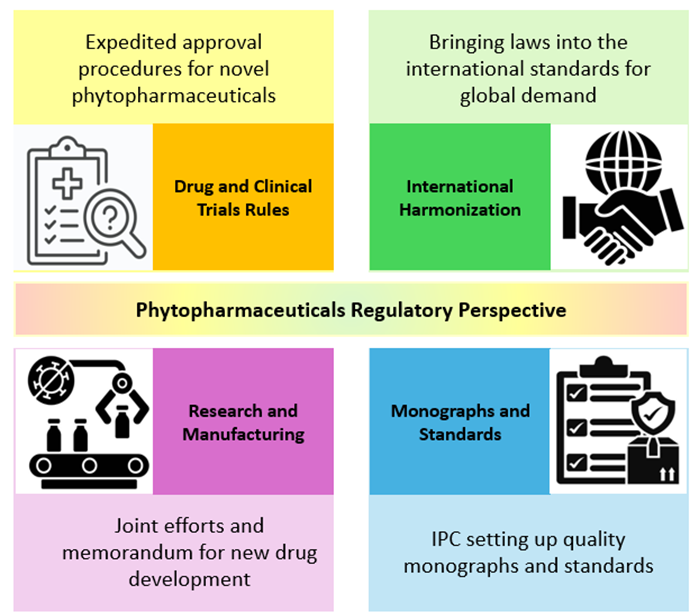
The [Fig. 1] summarizes the key pillars shaping the regulatory evolution of phytopharmaceuticals in India. It highlights four interconnected dimensions: expedited approval mechanisms under the Drugs and Clinical Trials Rules (2019), international harmonization efforts, strengthened research and manufacturing collaborations, and the development of pharmacopoeial monographs and quality standards by the Indian Pharmacopoeia Commission. Together, these components illustrate the structural transformation of India’s phytopharmaceutical framework from a traditionally rooted system toward a scientifically regulated and globally aligned drug development model.
The implementation of the New Drugs and Clinical Studies Rules (NDCTR), 2019, signified a significant structural enhancement by accelerating approval procedures and establishing a risk-based monitoring system for clinical studies. By specifying defined review periods and the enhancement of procedural transparency, NDCTR improved the predictability of the approval process for new therapies, including phytopharmaceuticals. Nevertheless, despite these operational efficiencies, the full advisory on phytopharmaceuticals particularly concerning botanical standardization, acceptable therapeutic objectives, and toxicological thresholds remains insufficiently generated. This gap sometimes delays starting trials, which can weaken trust from sponsors and reduce the number of applications[44].
The Government of India initiated the Phytopharmaceuticals Mission, coordinated by the Department of Biotechnology (DBT), to foster innovation in the development of plant-derived pharmaceuticals. The mission prioritizes the development of industry–academia partnerships, infrastructure strengthening, and translational research, particularly in regions that are abundant in biodiversity, such as Northeast India. Furthermore, collaborations such as the Memorandum of Understanding (MoU) between ICMR and Emami Ltd. for developing a phytopharmaceutical targeting pre-diabetes illustrate increasing public–private engagement within this regulatory framework[45, 46].
The Indian Pharmacopoeia Commission (IPC) has incorporated phytopharmaceutical monographs to standardize identity, purity, potency, and stability parameters[48, 49]. These standards are essential for ensuring reproducibility and regulatory compliance, particularly given the inherent variability of plant-based materials. In addition, the anticipated notification of CSIR-Indian Institute of Integrative Medicine (IIIM), Jammu, as a dedicated Drug Testing Laboratory (DTL) for phytopharmaceuticals signals efforts to strengthen regulatory infrastructure[49, 50].
India is also pursuing gradual alignment with international standards, including WHO guidelines and ICH principles, to enhance global acceptance and export competitiveness. However, unlike the FDA’s established Botanical Drug pathway or the European Union’s Traditional Herbal Medicinal Products Directive (THMPD), India’s phytopharmaceutical regulatory framework lacks comprehensive procedural guidance documents that clearly delineate evaluation criteria. This relative regulatory ambiguity may partially explain the cautious industry participation observed thus far[51, 52].
The current situation shows a regulatory framework that is in transition. It has official legislative approval, growing institutional support, and shorter timelines for procedures. Rectifying these structural deficiencies via more explicit assessment frameworks, improved harmonization, and focused incentives for clinical research would be crucial to realize India’s whole phytopharmaceutical potential.
India’s phytopharmaceutical market presents significant potential; however, regulatory segmentation between AYUSH products and CDSCO-regulated phytopharmaceuticals has created a dual-market structure. While companies such as Dabur, Himalaya, Patanjali, and Zandu dominate the herbal and AYUSH-based commercial segment, relatively few entities have pursued the phytopharmaceutical new drug pathway under CDSCO due to higher compliance costs and rigorous clinical requirements[53, 54].
This regulatory divide influences investment patterns, as firms often prefer the comparatively less stringent AYUSH route over the phytopharmaceutical drug pathway. Consequently, despite India’s biodiversity advantage, the number of CDSCO-approved phytopharmaceutical drugs remains limited. Strengthening regulatory clarity, providing incentives for clinical research, and aligning with international botanical drug frameworks may encourage greater industry participation in the phytopharmaceutical category.
Thus, regulatory structure directly shapes market behavior, innovation intensity, and global competitiveness of India’s plant-derived therapeutics sector. India’s phytopharmaceutical industry is expanding significantly, mostly due to rising consumer awareness of wellness and health issues and a noticeable trend towards natural and organic goods[55].
Several companies are at the forefront of the phytopharmaceutical market in India, each contributing to the sector's growth through innovative products and strong market presence:
Himalaya Drug Company: Renowned for its extensive range of herbal products, himalaya has established a robust presence in both domestic and international markets. The company focuses on quality and efficacy, leveraging traditional knowledge and modern science.
Dabur India Ltd.: Dabur, a leader in the herbal industry, provides a wide variety of goods, such as ayurvedic medications, personal care products, and health supplements. The company’s market position is improved by its well-known brand and robust distribution network.
Patanjali Ayurveda: This rapidly growing company has gained immense popularity for its focus on ayurvedic products, capturing a significant market share. Patanjali's marketing strategies and emphasis on natural ingredients resonate well with health-conscious consumers.
Zandu Pharmaceutical Works Ltd.: Offering a range of goods, including ayurvedic formulations, Zandu has a long history of using herbal therapy. The business is renowned for its traditional methods and dedication to quality.
Targeted regulatory refinement is essential to unlock the full potential of phytopharmaceuticals in India. The Central Drugs Standard Control Organization (CDSCO) is advised to issue a dedicated guidance document that delineates evaluation criteria specific to phytopharmaceuticals. This document should encompass expectations regarding botanical standardization, clinical endpoints, and toxicological assessments. Implementing a streamlined or adaptive approval pathway, while ensuring scientific rigor, has the potential to improve industry participation. Financial incentives, research grants, and models of public–private partnerships can enhance support for clinical development and translational research. Enhancing the capabilities of specified drug testing laboratories and broadening pharmacopoeial standards will lead to improved quality assurance and increased regulatory confidence. Proactive harmonization with international botanical drug frameworks, including alignment with WHO and ICH principles, will enhance global competitiveness and facilitate export opportunities. A systematic, policy-oriented strategy that combines regulatory clarity, institutional backing, and innovation incentives will be essential in evolving India’s phytopharmaceutical sector into a globally acknowledged evidence-based industry.
The evolving regulatory ecosystem for phyto- pharmaceuticals in India, highlighting key policy reforms, institutional initiatives, and standardization efforts that collectively shape the regulatory landscape. The introduction of the Drugs and Clinical Trials Rules, 2019 has streamlined and expedited the approval process for novel phytopharmaceuticals by establishing defined timelines and adopting a risk-based approach to clinical trial monitoring. At the same time, India is working toward international harmonization by aligning its regulatory requirements with global standards such as those of the WHO and ICH, which is essential for improving export potential and global competitiveness. Collaborative efforts between government agencies such as DBT, ICMR, and CSIR and industry partners are strengthening research capacity and supporting the establishment of modern manufacturing facilities for phytopharmaceutical production. Additionally, the Indian Pharmacopoeia Commission is developing monographs and quality standards to ensure the purity, potency, and stability of these products. Together, these measures reflect India’s transition toward a more structured, science-based, and globally aligned regulatory framework for phytopharmaceuticals.
1. Shrivastava SR, Shrivastava PS, Ramasamy J. Mainstreaming of Ayurveda, Yoga, Naturopathy, Unani, Siddha, and Homeopathy with the health care delivery system in India. Journal of Traditional and Complementary Medicine. 2015; 5 (2). Available from: https://doi.org/10.1016/j.jtcme.2014.11.002
2. Nooreen Z, Rai VK, Yadav NP. Phytopharmaceuticals: A new class of drug in India. Annals of Phytomedicine: An International Journal. 2018; 7 (1). Available from: https://doi.org/10.21276/ap.2018.7.1.4
3. Bhusnure OG, Shinde MC, Vijayendra SSM, Gholve SB, Giram PS, Birajdar MJ. Phytopharmaceuticals: An emerging platform for innovation and development of new drugs from botanicals. Journal of Drug Delivery and Therapeutics. 2019; 9 (3-s). Available from: https://doi.org/10.22270/jddt.v9i3-s.2940
4. Singh A, Kalaivani M, Chaudhary P, Srivastava S, Goyal RK, Gupta SK. Opportunities and Challenges in Development of Phytopharmaceutical Drug in India- A SWOT Analysis. Journal of Young Pharmacists. 2019; 11 (3). Available from: https://doi.org/10.5530/jyp.2019.11.66
5. Prajapati P, Pathan A, Prajapati D. Phytopharmaceuticals Regulatory requirements and licensing process. International Journal of Drug Regulatory Affairs. 2023; 11 (3). Available from: https://doi.org/10.22270/ijdra.v11i3.615
6. Katiyar CK. Phytopharmaceuticals: Unique Regulatory Category with Immense Potential for Medical Unmet Needs. Natural Medicines. 2019; Available from: https://doi.org/10.1201/9781315187853-12
7. Central Drugs Standard Control Organization Directorate General of Health Services Ministry of Health and Family Welfare Government of India. <I>Frequently asked questions (FAQs) on approval of new phytopharmaceutical drugs</I>. 2018. https://cdsco.gov.in/opencms/export/sites/CDSCO_WEB/Pdf-documents/New-Drugs/FAQs/New_Drugs_FAQs.doc.
8. Bhatt A. Phytopharmaceuticals: A new drug class regulated in India. Perspectives in Clinical Research. 2016; 7 (2). Available from: https://doi.org/10.4103/2229-3485.179435
9. Sharma S. Current status of herbal product: Regulatory overview. Journal of Pharmacy And Bioallied Sciences. 2015; 7 (4). Available from: https://doi.org/10.4103/0975-7406.168030
10. Nirmal P, Singh R, Kumar N, Sharma S. Phytopharmaceutical regulated new class: An Industrial initiative of Ayurvedic drugs towards the advancement of India system of medicine. World Journal of Advanced Research and Reviews. 2022; 15 (3). Available from: https://doi.org/10.30574/wjarr.2022.15.3.0949
11. Yarnell E. <I>Artemisia annua</I> (sweet annie), other artemisia species, artemisinin, artemisinin derivatives, and malaria. Journal of Restorative Medicine. 2014; 3 (1). Available from: https://doi.org/10.14200/jrm.2014.3.0105
12. Al-Kuraishy HM, Al-Gareeb AI, Naji MT, Al-Mamorry F. Role of vinpocetine in ischemic stroke and poststroke outcomes: A critical review. Brain Circulation. 2020; 6 (1). Available from: https://doi.org/10.4103/bc.bc_46_19
13. Salehi B, Staniak M, Czopek K, Stępień A, Dua K, Wadhwa R, <I>et al</I>. The Therapeutic Potential of the Labdane Diterpenoid Forskolin. Applied Sciences. 2019; 9 (19). Available from: https://doi.org/10.3390/app9194089
14. Arul Kumar R, Indrapriyadharshini C, Parthiban S, Boopathi T, Alexander J. Review on properties of <I>Boswellia serrata</I> in inflammatory and rheumatoid arthritis (RA) management. World Journal of Pharmaceutical Research. 2022; 11 (9). Available from: https://doi.org/10.20959/wjpr20229-24509
15. Nash KM, Shah ZA. Current Perspectives on the Beneficial Role of <i>Ginkgo biloba</i> in Neurological and Cerebrovascular Disorders. Integrative Medicine Insights. 2015; 10 Available from: https://doi.org/10.4137/imi.s25054
16. Gachelin G, Garner P, Ferroni E, Tröhler U, Chalmers I. Evaluating <i>Cinchona</i> bark and quinine for treating and preventing malaria. Journal of the Royal Society of Medicine. 2017; 110 (2). Available from: https://doi.org/10.1177/0141076816688411
17. Csiszar A. Anti‐inflammatory effects of resveratrol: possible role in prevention of age‐related cardiovascular disease. Annals of the New York Academy of Sciences. 2011; 1215 (1). Available from: https://doi.org/10.1111/j.1749-6632.2010.05848.x
18. Abascal K, Yarnell E. Increasing Vitality with Adaptogens: Multifaceted Herbs for Treating Physical and Mental Stress. Alternative and Complementary Therapies. 2003; 9 (2). Available from: https://doi.org/10.1089/107628003321536959
19. Al meleebia TM, Alsayari A, Wahab S. Pharmacological and Clinical Efficacy of Picrorhiza kurroa and Its Secondary Metabolites: A Comprehensive Review. Molecules. 2022; 27 (23). Available from: https://doi.org/10.3390/molecules27238316
20. Hossain CM, Gera ME, Ali KA. Current status and challenges of herbal drug development and regulatory aspect: a global perspective. Asian Journal of Pharmaceutical and Clinical Research. 2022; 15 Available from: https://doi.org/10.22159/ajpcr.2022.v15i12.46134
21. Mukherjee PK, Bahadur S, Chaudhary SK, Kar A, Mukherjee K. Quality Related Safety Issue-Evidence-Based Validation of Herbal Medicine Farm to Pharma. Evidence-Based Validation of Herbal Medicine. 2015; Available from: https://doi.org/10.1016/b978-0-12-800874-4.00001-5
22. Prabhakar P, Mamoni B. Technical problems, regulatory and market challenges in bringing herbal drug into mainstream of modern medicinal practices. <I>Research Journal of Biotechnology</I> 2021;16:3.
23. Wu C, Lee SL, Taylor C, Li J, Chan YM, Agarwal R, <I>et al</I>. Scientific and Regulatory Approach to Botanical Drug Development: A U.S. FDA Perspective. Journal of Natural Products. 2020; 83 (2). Available from: https://doi.org/10.1021/acs.jnatprod.9b00949
24. CDSCO. <I>Frequently Asked Questions (FAQs) on Approval of new Phytopharmaceutical Drugs</I>. 2022 Nov. Available from: https://cdsco.gov.in/opencms/export/sites/CDSCO_WEB/Pdf-documents/New-Drugs/FAQs/New_Drugs_FAQs.doc
25. Jadhav R, Das J, Rajyaguru S, Kyada S, Kale V, Teja PK, <I>et al</I>. Regulations, current development, and future prospects of phytopharmaceuticals, a new class of herbal medicines in India. Discover Pharmaceutical Sciences. 2025; 1 (1). Available from: https://doi.org/10.1007/s44395-025-00006-4
26. Ministry of AYUSH, Government of India. <I>Guidelines for Phytopharmaceutical Drug Development in India</I>. Ministry of AYUSH, India, 2016. Available from: https://pharmaceuticals.gov.in/sites/default/files/phytopharmaceutical_guidelines.pdf
27. Chaudhary K, Singh I. Regulatory Requirements for Phytopharmaceuticals Under The Central Drug Standards Control Organization (CDSCO). <I>Research Journal of Phytochemistry</I>. 2022; 16(1): 23. https://rjp.scione.com/cms/abstract.php?id=32
28. <I>Ministry of Health and Family Welfare Gazette Notification G.S.R. 918(E)</I>. Available from: http://www.cdsco.nic.in/writereaddata/GSR%20918-E-dated-30-11-2015.pdf .
29. Singh N, Madkaikar NJ, Gokhale PM, Parmar DV. New drugs and clinical trials rules 2019: Changes in responsibilities of the ethics committee. Perspectives in Clinical Research. 2020; 11 (1). Available from: https://doi.org/10.4103/picr.picr_208_19
30. Jadaun GPS, Rastogi S, Kumar A, Chauhan J. Ensuring the quality of medicines in India: An update on the development, modernization, and harmonization of drug standards in the Indian Pharmacopoeia. Saudi Pharmaceutical Journal. 2023; 31 (12). Available from: https://doi.org/10.1016/j.jsps.2023.101825
31. Ravi G, Deepa K, Ezhilarasi V. Quality Control and Standardization of Herbal Drugs and Phytopharmaceuticals. <I>Journal of Research in Pharmaceutical Science</I>, 12(5):281-287, 2021.
32. Verma N, Pingili D, Awasthi A; Regulatory Landscape For Phytopharmaceuticals In India; <I>International Journal of Pharmacy and Pharmaceutical Sciences</I>. (2025) 15 (1): 27-29.
33. CSIR - Indian Institute of Integrative Medicine (IIIM). Phytopharmaceuticals Mission: <I>Medicinal plants have played a key role in human health since time immemorial</I>. 2021. https://iiim.res.in/phytopharmaceuticals-mission/.
34. <I>Phytopharmaceutical Drug; Cliniexperts</I>; Available from: https://cliniexperts.com/india-regulatory-services/phytopharmaceutical-drug/
35. <I>Introduction and importance of medicinal plants and herbs</I>. National Health Portal (NHP), India. 2022. Available from: https://www.nhp.gov.in/introduction-and-importance-of-medicinal-plants-and-herbs_mtl
36. <I>WHO guidelines for assessing quality of herbal medicines with reference to contaminants and residues</I>; 2007. Available from: https://www.who.int/publications/i/item/9789241594448
37. <I>ICH Guidelines</I>, Available from: https://www.ich.org/page/ich-guidelines
38. Kaushik R, Sharma P; From Tradition to Global Standards: Governance, Regulatory Challenges and Strategic Reforms in Ayush Drug Regulation; <I>Traditional Medicine Review</I>; Vol. 5(2), 26-38 October 2025.
39. <I>Regulatory Challenges of Indian Drugs</I>; 2023. Available from: https://www.drishtiias.com/daily-updates/daily-news-editorials/regulatory-challenges-of-indian-drugs
40. Liu L, Wang Y, Zhang J, Wang S. Advances in the chemical constituents and chemical analysis of Ginkgo biloba leaf, extract, and phytopharmaceuticals. Journal of Pharmaceutical and Biomedical Analysis. 2021; 193 Available from: https://doi.org/10.1016/j.jpba.2020.113704
41. Meshnick S. Artemisinin: mechanisms of action, resistance and toxicity. International Journal for Parasitology. 2002; 32 (13). Available from: https://doi.org/10.1016/s0020-7519(02)00194-7
42. Pavithra K, Manimaran V. A Review of Safety, Quality, Regulation, and Delivery Approaches for Phytopharmaceuticals. Jordan Journal of Pharmaceutical Sciences. 2024; 17 (2). Available from: https://doi.org/10.35516/jjps.v17i2.1768
43. Maheshbhai, PM, Asmatbanu P. Current Regulatory Aspects of Phytopharmaceutical in India and Europe. <I>Pharmaceutical Regulatory Affairs</I> 11 (2022): 303
44. <I>New Drugs and Clinical Trials Rules</I>, 2019. Available from: https://www.cdsco.gov.in/opencms/opencms/en/Acts-and-rules/New-Drugs/
45. <I>Phytopharmaceuticals Mission : Medicinal plants have played a key role in human health since time immemorial</I>; Council of Scientific & Industrial Research; Available from: https://iiim.res.in/phytopharmaceuticals-mission/
46. ETHealthWorld; <I>ICMR, Emami sign agreements to develop herbal drugs</I>; 2017. Available from: https://health.economictimes.indiatimes.com/news/industry/icmr-emami-sign-agreements-to-develop-herbal-drugs/58840482
47. Kalaiselvan V, Tiwari R, Anantha Narayana DB, Prakash J; Phytopharmaceuitcals Ingredients’ – A new category of Monographs in Indian Pharmacopoeia; <I>Research & Reviews: Journal of Pharmacognosy & Phytochemistry</I>; 2021: Volume 9 (3). Available from: https://www.rroij.com/open-access/phytopharmaceuitcals-ingredients--a-new-category-ofmonographs-in-indian-pharmacopoeia.pdf
48. <I>Phytopharmaceuticals</I>; 2019; Available from: https://ipc.gov.in/about/us/departments/phytopharmaceuticals.html
49. Katiyar CK, Mandal SC, <I>et al</I>. <I>Newsletter. Society for Ethnopharmacology, INDIA</I>; Volume 2020/3 No. 22. Available from: https://ethnopharmacology.in/wp-content/uploads/2020/10/Newsletter-22.pdf
50. Gireesh B. <I>Two phytopharmaceutical leads from CSIR-DBT-ICMR collaboration in the stage of IND-enabling studies</I>; Pharmabiz; 2023. Available from: https://www.pharmabiz.com/NewsDetails.aspx?aid=165040&sid=1
51. Suvarna MB, Smita T, Pooja K, Sangeeta J, Shreya K, Alqama K; WHO & ICH Guidelines for the Assessment of Herbal Drug. International Journal of Trend in Scientific Research and Development (ijtsrd); 2022 April; 6(3); pp.549-559, Available from: www.ijtsrd.com/papers/ijtsrd49571.pdf
52. Shah S; <I>Phytopharmaceutical Drugs and Their Related Regulations in India</I>; 2018. Available from: https://cliniexperts.com/phytopharmaceutical-drugs-and-their-related-regulations-in-india/
53. Madhavi BLR, Pruthvi N, Chandur U, Sundari SP; <I>Phytopharmaceutical industry in India: An insight towards its growth and sustenance</I>; APTI Women’s Forum Newsletter, Sept - Dec, 2024, 215-219.
54. Sahoo B, Roy A, Vishwakarma M, Soni V; <I>Phytopharmaceuticals in India: A Comprehensive SWOT Analysis and Market Overview</I>; APTI Women’s Forum Newsletter, Sept - Dec, 2024, 67-74
55. Smarta RB. Phytopharmaceutical and emerging opportunities in indian pharma market health vision; 2022. Available in https://healthvision.in/phytopharmaceutical-and-emerging-opportunities-in-indian-pharma-market/
© 2026 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.