

Journal of Pharmaceutical Research
Year: 2024, Volume: 23, Issue: 4, Pages: 242-245
Original Article
Shailesh B Patil1,∗, Jitendra D More2
1Department of Pharmaceutical Chemistry, DCS’s ARA College of Pharmacy, Nagaon, Dhule, University of KBC-NMU, Jalgaon, Maharashtra, India
2Department of Pharmaceutical Chemistry, Ravindra Nikam College of Pharmacy, Gondur, Dhule, University of KBC-NMU, Jalgaon, Maharashtra, India
*Corresponding Author
Email: [email protected]
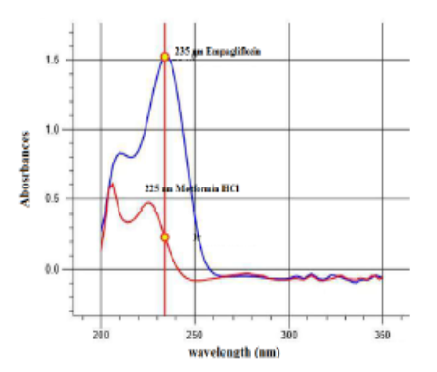
A validated UV spectrophotometric method has been developed for the simultaneous quantification of Gibtulio Met (12.5/500mg) Fixed Dose Combination dosage forms using the simultaneous equation method. This approach is based on measuring the absorbance of Metformin HCl and Empagliflozin within the concentration ranges of 2-10 μg/ml and 0.1-5 μg/ml, respectively, at their respective λ max values of 235 nm and 225 nm. The linearity correlation coefficients for Metformin HCl and Empagliflozin in these concentration ranges were 0.999, and 0.9998 respectively. The results were statistically validated in compliance with ICH guidelines.
Keywords: Metformin, Empagliflozin, ICH guidelines
© 2024 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.