

Journal of Pharmaceutical Research
Year: 2025, Volume: 24, Issue: 4, Pages: 231-238
Original Article
Maryam1, Muhammad Muazzam Nasser2, Muhammad Anjum Zia3*, Fatima Idrees3
1School of Chemistry, Xi'an Jiaotong University, China
2Institute of Biomedical and Health Engineering, Xi’an Jiaotong University, China
3Department University at University of Agriculture, Faisalabad, Pakistan
*Corresponding author.
Muhammad Anjum Zia
Email: [email protected]
A type of illness known as cardiovascular disease affects the heart or blood arteries. The flow of blood to the heart, brain, or body is reduced due to thrombosis (blood clotting). Streptokinase (SK) is an extracellular enzyme that converts plasminogen to plasmin, as a medication in thrombolytic treatment. The current study was focused on streptokinase production and enhanced activity using magnetic nanoparticles. Nutrient agar media and liquid state fermentation at pre-optimized conditions was used. The proteolytic activity of the enzyme was determined by using the casein digestion method and then Biuret test was performed for protein estimation. The product of bacterial culture was extracted, and the partial purification of streptokinase was done by ammonium sulfate precipitation, dialysis and then further chromatographic techniques and gel filtration were applied. Immobilization of streptokinase was done by using Fe3O4 magnetic nanoparticles to enhance its activity and protein yield. The immobilization of SK on magnetic nanoparticles was characterized by UV-Vis spectroscopy and Zetasizer. Fe3O4 magnetic nanoparticle showed absorption peak at 224.4 nm and size of 229.4 nm. When compared to the total activity of gel chromatography (5.10 U/ml), immobilization increased the total activity of SK to 7.106 U/ml. In this way enzyme activity of immobilized streptokinase increased by 1.39-fold. Immobilized enzyme was used in in vivo studies and then compared with native one. The activity analysis of immobilized streptokinase was compared with the native one. Immobilized streptokinase exhibited more thrombolytic activity in rabbits than the native form, as demonstrated by in vivo study.
Keywords: Streptokinase; Immobilized enzyme; Nanoparticle; Chromatography; Streptococcus pyogenes; Thrombosis
Impaired oxygen delivery to cardiac tissues or the formation of intravascular clots can result in severe outcomes such as coronary artery disease, pulmonary embolism, or sudden cardiac arrest [1]. The primary global causes of death are coronary heart illnesses, which include myocardial infarction, stroke, embolism, and other heart disorders [2]. Thrombosis-related conditions such as thrombophlebitis, phlebitis, and venous thrombosis contribute to the development of embolic disorders. Several anticoagulants, such as heparin, are currently utilized to treat thrombosis disorders despite the potential for a brain hemorrhage. Plasminogen activators used in thrombolytic therapy have been proposed as a better treatment option than other anticoagulants [3]. Apart from surgical operations to eliminate or bypass the clot, or the formation of deposit arteries to offer an alternative source of blood, only therapy option is the injection of clot busters medicines such as streptokinase to destroy the blood clot [4]. In 1933, William Smith developed streptokinase (SK) initially, known as fibrinolysin because of its role in the breakdown of fibrin. Streptococcus bacteria produce mature protein SK, a 414-amino acid, 47-kDa single-chain protein without disulfide bridges [5]. Because of its low cost and powerful therapeutic effects, It is the one of these that is most frequently utilized in underdeveloped nations [6]. Despite its clinical utility, streptokinase suffers from a short circulation half-life and is associated with adverse effects including chills, fever, vomiting, gastrointestinal bleeding, and immune-related reactions [7].
Currently, biotechnology companies worldwide are searching for methods to boost the efficiency of enzymes and develop novel approaches to prolong their shelf life. Compared to free enzymes in solution, immobilized enzymes are more resilient and long-lasting to changes in the environment. More importantly, a wider variety of bioreactor designs, simpler enzyme and product recovery, various enzyme uses, continuous enzymatic process operation, and quick reaction termination are made possible by the heterogeneity of the immobilized enzyme systems [8]. It has been created expressly to severely impede an enzyme's or cell's freedom of movement [9].
Nanoparticle utilization has increased rapidly across many different companies. They serve as a key tool in the detection and treatment of diseases by enhancing the effects of bioactive compounds via immobilization on solid supports, encapsulation, and biological tissues [10]. The magnetic mineral Fe3O4, because of its biocompatibility and greater magnetization, is generally served as a support substance for enzyme. Fe3O4 magnetic nanoparticles, Streptokinase was rendered immobile by coating it with polyaniline [11]. The MNPs' immobilized enzymes demonstrated a greater temperature and pH operating range, along with improved thermal and storage stabilities, as compared to their pristine equivalents [12]. Immobilization evolved to increase the enzyme's activity. After being immobilized on the magnetic nanoparticle (MNP), the fibrinolytic streptokinase was evaluated for its thrombolytic and streptokinase properties [13]. Streptokinase is frequently utilized in in vivo thrombolysis therapy because of its efficacy as a thrombolytic medication [14]. In animal embolus models, the thrombolytic drugs immobilized on magnetic nanoparticles (MNPs) showed stability and an efficient fibrinolytic therapy in comparison to native therapies [15].
The streptokinase enzyme immobilized on Fe3O4 was described as an effective nano-biocatalyst in the current study. Subsequently, the effects of administering immobilized streptokinase on the lysis of rabbit fibrin clots were investigated.
The experiment was carried out at the University of Agriculture in Faisalabad, Pakistan, at the Enzyme Biotechnology Laboratory inside the Department of Biochemistry. The study's foundation was the Streptococcus pyogenes EBL-48 strain, which was acquired from the Enzyme Biotechnology Laboratory. The streptokinase enzyme was produced by the Streptococcus pyogenes EBL-48 strain that had been previously optimized. The generated streptokinase was subsequently purified using a variety of chromatography methods, including gel filtration and ion exchange.
On the nutrient agar media, Streptococcus pyogenes EBL-48 bacteria were cultivated. Nutrient agar plates were used to support the bacterial culture. Using a digital weight balance, all the components (Agar: 2.8 g, peptone: 1 g, yeast extract: 0.5g, NaCl: 0.5 g, distilled water: 100 ml) were measured to create the nutritional agar media at 6.8-7.4 pH. Streptokinase production was carried out following the method described by Zhang (2023). Further, For the inoculum preparation all the chemicals of liquid broth media (peptone: 1 g, NaCl: 0.5 g, yeast extract: 0.5 g, Distilled water: 100 ml) weighed with the help of weighing balance and then dissolved in a flask containing 100 milliliters of distilled water. The medium was then sterilized at 121°C for 15 minutes. After that bacterial culture was injected into the flask then placed in an orbital shaker for 24 hours at 37°C and 120 rpm by using the previous method [16].
The fermentation media was made by combining all the components (KH2PO4: 0.15 g, CaCO3: 1.5 g, glucose: 3 g, yeast: 1.8 g, sugar cane: 1.2 g,) with 100 ml of distilled water at 6 pH under optimized conditions by using the previous method [17].
Casein digestion assay was used to analyze streptokinase. Tyrosine, along with other amino acids and peptide fragments, are released during enzyme digestion of casein upon plasminogen activation. By mixing 50 mM Tris HCl (pH 8.0) with 10 mg of casein in 0.1 ml of sample solution, a mixture of around 2 ml was created. Then set aside for about 20 minutes of incubation at 37°C. 0.4 ml of 3.3M HCI was added to the mixture above after incubation. An extra 2.6 ml of 5% w/v trichloroacetic acid (TCA) was added to stop the reaction. After cooling on ice for approximately half an hour, the mixture was filtered using Whatman no. 1 filter paper (pore size ~11 µm) and analyzed using a spectrophotometer The enzyme activity was measured using the absorbance measured at 280 nm by using the method with some modifications by using previously method [18].
A substance known as biuret is produced by heating urea to 180°C. A chemical test known as the "Biuret test" employs the Biuret reagents, which include Copper II sulphate (CuSO4) in a 1% solution, in accordance with the previously described procedure [19]. Primarily, collected test tubes with the labels "blank test tube" and "sample test tube." Enzyme sample was in the sample test tube, which also held distilled water as a blank. The test tubes were then filled with 1 ml of the Biuret reagent, which was added, and incubated at 37°C for 20 minutes. A difference in color was noticed. This solution was turned into a purple colour. After that, the sample was analyzed using a spectrophotometer to measure its absorbance at 540 nm by using a previously method [20].
The produced enzyme was then purified using a variety of techniques, such as ammonium sulphate precipitation, ion-exchange chromatography, and gel-filtration chromatography. Partial purification was done by Ammonium sulphate precipitation (Salting In & Salting Out), Dialysis, ion exchange (DEAE cellulose) chromatography, and Gel (sephadexG-150) chromatography was used by previously method [5].
Added 50 mL of distilled water to a solution containing 5 mL of 1.1 M FeCl3 and 5 mL of 0.6 M FeSO4 then placed it on the magnetic stirrer at 25°C.To precipitate the particles, dropwise additions of NaOH (5.0 M) were made till pH 10. Then placed on water bath at 50°C for 30 minutes by continuous stirring. Continuously washed with water to neutralize the nanoparticles. After that placed these nanoparticles in oven for about 2 hours at 50°C. Weighed the particles. By introducing 0.5 g of magnetic nanoparticles to 50 mL of KMnO4 solution (0.1 M) at 25°C for 1 hour while gently magnetic stirring, magnetic nanoparticles were coated. Following this, the nanoparticles were repeatedly washed with distilled water until they became transparent. Next, 50 mL of a 0.5 M aniline solution that had been solubilized with 1.0 M HNO3 was used to dissolve the magnetic KMnO4 nanoparticles. With mild magnetic stirring, polymerization occurred at 4°C for an hour. The magnetic nanoparticles coated with polyaniline were rinsed several times using distilled water, and 0.1 M citric acid. Furthermore, using a previously described technique, they were dried at 50°C in a drying oven and stored at ambient temperature (around 25°C) until needed in an airtight container [21].
Nevertheless, it had been altered to render the Streptokinase immobile. Nanoparticles (10 mg/mL) were mixed with 2% glutaraldehyde in microtubes. For four hours, the material was mechanically agitated while being kept out of the light.
After three rinses with 245 mM sodium phosphate buffer (pH 7.2), the particles were rinsed ten times with distilled water. Each microtube was filled with 1 mL of SK (5 mg/ml), and the mixture was mechanically agitated for two hours. Following two washes of the nanoparticles with the buffer, 1.0 mL of 0.1 M glycine was added to inhibit any residual reactive groups. After that, the mixture was stirred for 30 minutes in a cold chamber. Using a previously described technique, the magnetic nanoparticles containing the SK/MNPs were subsequently washed with sodium phosphate solution and stored at room temperature in order to ascertain the activities later [13, 22].
The suspensions (1 ml) were mixed with 0.2 M Tris-HCl, pH 7.2, 103 M CaCl2, 1.0% (w/w) casein, and 1 mg of the (MNP/SK), and they were then incubated for 1 hour at 28°C.The reaction was then stopped by adding 1.0 mL of a 10% (w/v) trichloroacetic acid solution. After centrifugation at 3000 g for 15 minutes, an aliquot (0.8 mL) of the supernatant was transferred to a second tube that contained 0.2 mL of 1.8 M NaOH. After being homogenized, the samples were examined using UV-Vis spectrophotometry at a wavelength of 420 nm. One unit of streptokinase activity is the amount of enzyme that leads to a 0.01 increase in absorbance per minute.
In vivo experiment three male adult rabbits were used. All animal procedures were approved by the Institutional Animal Ethics Committee of the University of Agriculture, Faisalabad, at Department of Biochemistry (letter No. 479) and were conducted in accordance with national guidelines. To anesthetize the animals about 1ml of lignocaine was injected into the site of surgery. After that it was surgically cut to expose the jugular vein of rabbit. Then this exposed vein was treated with a few drops of FeCl3 saturated filter paper for 3-5 minutes. After the formation of blood clots, the animal which received 1 ml IV heparin (5000 U) was regarded as a negative control group. 1m of Streptokinase produced and purified in EBL has injected IV of 2nd animal. The third was injected IV with 1ml of immobilized streptokinase produced and purified in EBL. After 1 hour, Blood flow velocity in the jugular vein was measured by ultrasound (prob 7 MHz) to confirm lysis of induced thrombosis. After that, observed blood flow movement on ultrasonography. The speed of the blood in the vessel affects the difference in frequency between ultrasonic waves that are transmitted and reflected by using the previous method [23].
All experiments were performed in triplicate (n = 3). Data were analyzed using SPSS software. Statistical comparisons between groups were made using one-way ANOVA. A p-value < 0.05 was considered statistically significant.
Plenty of bacteria, including Bacillus, Pseudomonas, Staphylococcus, Alteromonas, Coryne, and Escherichia coli, are known to produce streptokinase. According to reports, S. equisimilis is a reliable source of SK among the Streptococci from human hosts [24]. On nutritional agar media, the Streptococcus pyogenes strain used in this experiment was maintained. Streptococcus pyogenes EBL-48 was cultivated using 0.8 g sugarcane bagasse as a substrate for streptokinase production under liquid-state fermentation at 37°C, 120 rpm, and pH 7 for 24 hours and then filtered with Whatman no.1 filter paper (pore size ~11 µm) to remove cell debris.
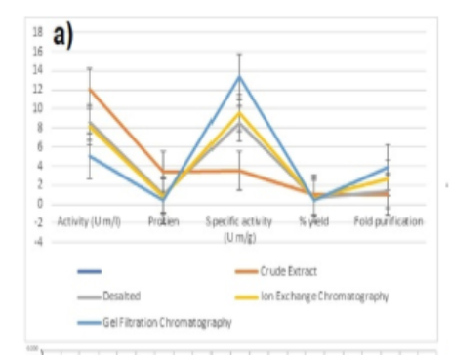
Increase of protein purity is a serious challenge in the production of recombinant therapeutic proteins. Chromatographic techniques were carried out for the purification of streptokinase. Various steps of chromatographic techniques were carried out for the purification of the enzyme. In [Table. 1] and [Table. 2], the analysis of the variation following each phase (crude enzyme extract, dialysis, ion exchange, and gel filtration) are provided. Streptokinase activity showed a decreasing trend across purification steps: from 12.12 U/mg in the crude extract to 5.10 U/mg after gel filtration [Fig. 1]a. This work correlates with more recent work [25]. According to [Table. 1], the enzyme's protein content gradually decreases. The protein content value of crude enzyme extract is 3.46 mg/ml, 1.01 mg/ml after dialysis, 0.85 mg/ml after ion exchange and 0.38 mg/ml after gel filtration showing significant statistical results of one way ANOVA described in [Table. 2].
The probability value for the protein content is (P<0.05), indicating that our results from the current study are statistically highly significant and are consistent with those of another study [26]. It was evident from a drop in protein levels that the enzyme functions more effectively after purification. After purification, streptokinase dissolves blood clots more quickly than the enzyme in its raw form. Ghosal also came to the conclusion that the specific activity of the enzyme rises with each step. More recently, according to researchers [27], the specific activity was 127 U/mg after ion exchange and 18.82 U/mg after size exclusion chromatography. [Table. 2] displays the statistical outcome of the specific streptokinase activity. Our research's findings have been found to be significant because the statistical analysis’s probability value is less than 0.05. To produce high-quality streptokinase-specific activity, the U/mg enzyme count must be raised throughout the purification process. The specific activity increasing from left to right is seen in [Fig. 1]a. This means that the specific activity after gel filtration is higher than all previous ones. This pattern is also observed in the research [28]. The specific activity of crude enzyme extract is 3.50 U/mg, desalted is 8.50 U/mg, after ion exchange it is 9.62 U/mg and after gel filtration, it is 13.42 U/mg. The frequency of enzyme purification increases from first to last after purification using various methods. The statistical analysis of streptokinase following multiple purifications is shown in [Table. 2], and it came to the conclusion that the results are significant (P< 0.05). The purification factor obtained after crude enzyme extract is 01, the purification factor after ammonium sulphate precipitation is 1.43, the purification factor after ion exchange is 2.75, and the purification factor after gel filtration is 3.84.
| Purification step | Activity (U m/l) | Protein (mg m/l) | Specific activity (U m/g) | % yield | Fold purific-ation |
|---|---|---|---|---|---|
| Crude Extract | 12.12 | 3.46 | 3.50 | 100% | 1 |
| Desalted | 8.59 | 1.01 | 8.50 | 70.87% | 1.43 |
| Ion Exchange Chromatography | 8.18 | 0.85 | 9.62 | 67.49% | 2.75 |
| Gel Filtration Chromatography | 5.10 | 0.38 | 13.42 | 42.08% | 3.84 |
The purity factor increases after each cleaning stage. The present study is consistent with Narasimhan's [29] purification of the enzyme using the same sequence, which produced purifications of 01, 2.6, 4.9, and 10.1, respectively. After each phase of purification, the yield of enzymes declines in percentage. According to [Table. 1], the crude enzyme extract recovery percentage in the current study is 100%, which is ideal; following ammonium sulphate precipitation, it is 70.87%; for ion exchange, it is 67.49%; and for filtering in gel, it is 42.08%. The probability value in [Table. 2] is 0.000, which is less than 0.05 and demonstrates that the results of the percent yield analysis are significant. The new findings were comparable to Dabaa's study, which purified the enzyme and yielded percentage recoveries of 100% of the crude enzyme extract, 90% after ammonium sulphate, 74% after ion exchange, and 64% after gel filtration [5]. [Table. 1] provides a detailed explanation of the purification process for streptokinase. The purification steps led to a progressive increase in specific activity, from 3.50 U/mg in the crude extract to 13.42 U/mg after gel filtration, as summarized in [Table. 1]. Although total activity decreased from 12.12 U/ml
| Sum of Squares | Df | Mean Square | F-value | Sig. | ||
|---|---|---|---|---|---|---|
| Activity | Between Groups | 74.7578 | 3 | 24.9193 | 1383.76 | 0.000 |
| Within Groups | 0.1441 | 8 | 0.0180 | |||
| Total | 74.9019 | 11 | ||||
| Protein Content | Between Groups | 17.3497 | 6 | 2.89162 | 38338.43 | 0.000 |
| Within Groups | 0.0038 | 5 | 0.00075 | |||
| Total | 17.3535 | 11 | ||||
| Specific Activity | Between Groups | 150.436 | 4 | 37.6901 | 52131.44 | 0.000 |
| Within Groups | 0.005 | 7 | 0.0007 | |||
| Total | 150.441 | 11 | ||||
| Percentage Yield | Between Groups | 5103.30 | 5 | 1020.66 | 47240.59 | 0.000 |
| Within Groups | 0.13 | 6 | 0.02 | |||
| Total | 5103.43 | 11 | ||||
| Folds | Between Groups | 14.9088 | 4 | 3.72720 | 1665.34 | 0.000 |
| Within Groups | 0.0157 | 7 | 0.0024 | |||
| Total | 14.9245 | 11 |
NS= Non-significant (P>0.05), *=Significant (P>0.05), **=highly significant (P>0.05)
to 5.10 U/ml, this reduction was expected due to protein loss during each purification step. The overall purification fold achieved was 3.84. A summary of the results of the streptokinase analysis following purification procedures are
explained in more detail in [Fig. 1]a, which is a graphical depiction of this [Table. 1].
UV visible Spectroscopy is a potent analytical technique because it is frequently employed for analysis and because it may be used to partially detect functional groups in an organic chemical This spectroscopy primarily focuses on the absorption of electromagnetic radiation in the 200–800 nm range by any compound and identifying transitions or changes in the spectra of specific compounds that result from the excitation of electrons to higher energy levels in order to be able to verify the occurrences in this context. Chromophores are known for their chemical compounds' ability to absorb UV-Vis rays [30].
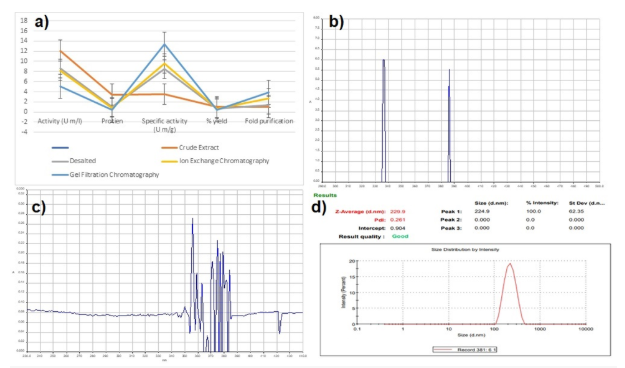
Fe3O4 magnetic nanoparticles were created in the current study using co-precipitation. By adding a base solution to a solution containing the iron ions, the co-precipitation method involves the simultaneous precipitation of iron ions and ions. In this investigation, sodium hydroxide served as the base solution. Two different iron salts, ferric chloride and ferric sulphate, were employed in the synthesis to compare their effects on the properties of the resulting MNPs. A constant molar ratio of 1:2 was maintained between the iron salt and the base solution [31]. The produced MNPs were characterized using UV-spectrophotometric methods from Hitech Lab, University of Agriculture Faisalabad, at wavelengths between 290 nm and 440 nm. The outcomes are consistent with the work [32], which demonstrates to the production of magnetic nanoparticles of Fe3O4.
Iron oxide nanoparticles have many advantages over other types of nanoparticles, such as biocompatibility, low toxicity, and magnetic properties that make it easy to remove them from the bloodstream [33]. Streptokinase (5 mg/mL), which had been purified by gel filtration chromatography, was added to the functionalized iron oxide MNPs together with the crosslinking agent glutaraldehyde. This made it easier for the streptokinase molecules to immobilize it on the surface of the nanoparticles by forming covalent connections with the iron oxide MNPs.
Immobilization makes the streptokinase more stable and active, which boosts its effectiveness in medicinal settings. Glutaraldehyde can stop streptokinase from leaking from the nanoparticles, which could reduce the efficacy of the treatment. Glycine was also added to the mixture to prevent nonspecific binding of streptokinase [34]. The absorbance peak verified the existence of streptokinase-immobilized magnetic nanoparticles and indicated that they are located between 340 nm and 440 nm [35].
The particle size can be estimated by measuring the variation in light intensity scattered by a suspension or solution. This technique is called dynamic light scattering, or DLS. The homogeneity and stability of nanoparticles are determined by their size and poly dispersion index, which also determines what will happen to them once they enter the human body. DLS was utilized to evaluate formulations that contained SK. The particle size of streptokinase immobilized nanoparticles was measured using a zeta-sizer (Z-Average nm) in order to examine the impact of adding enzyme on particle size, which correlates with [36]. [Fig. 1](d) showed the particle size of immobilized streptokinase about 244.9 ± 62.35 nm. A PDI of 0.261 indicates moderate monodispersity, suggesting a relatively uniform nanoparticle size distribution. The intercept value in DLS reflects the baseline signal; a value of 0.904 indicates a good correlation fit.
Numerous studies have been conducted to develop a strategy for thrombus induction in animal models [37], evaluated the in vivo activity of streptokinase on a rabbit model, so a rabbit was prepared for an in vivo investigation. For instance, Ferric chloride ions were employed to mimic the blood clot [38] created in the carotid artery of rats by inducing blood clots in the jugular veins of rabbits. In this study, thrombus was induced in the jugular vein using 20% FeCl₃ solution. Transamin was also used to create a thrombus in a rabbit’s carotids artery. One hour after the medicine was administered, the rate of thrombolysis was assessed. Determining thrombolysis in vivo is challenging since direct monitoring of plaque and thrombus is constrained.
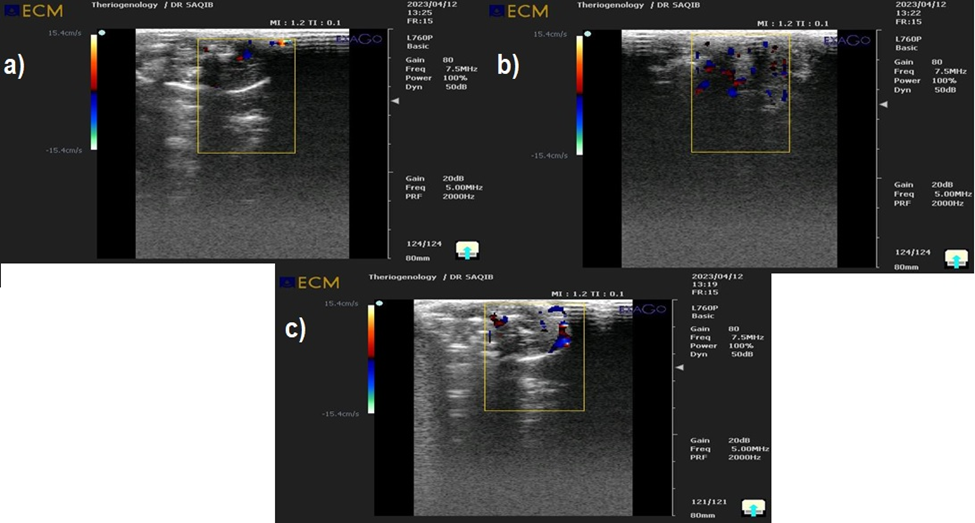
Using ultrasonography, the specific location of thrombus formation in the rabbit model was found. This work used ultrasonography to identify the precise location of the thrombus and establish a link between the thickness of the aortic stenosis and thrombus formation, as described in [Fig. 2] (a), (b), and (c). Our results demonstrate that the blood clot-induced disintegration was brought on by the streptokinase injection. When compared with the samples of Immobilized streptokinase by Fe3O4 magnetic nanoparticles and native streptokinase with heparin control, the results showed that immobilized streptokinase (streptococcus pyogenes EBL-48) by Fe3O4 magnetic nanoparticles completely lysed the induced clot while native streptokinase without streptokinase partially lysed the induced clot. According to these findings, the infusion of 4000 IU of streptokinase caused the extensive systemic activation of the fibrinolytic system.
Streptokinase produced from Streptococcus pyogenes EBL-48 was successfully purified and immobilized onto Fe₃O₄ magnetic nanoparticles. Immobilization enhanced the enzyme’s efficiency by 1.39-fold (measured at pH 7.4 and 37 °C), and the formulation remained stable under test conditions. No hemolysis was observed at any tested concentration, as determined by spectrophotometric analysis of hemoglobin release at 540 nm.
Further studies are recommended to evaluate the long-term stability, pharmacokinetics, and in vivo bio-distribution of immobilized streptokinase. In addition, the reusability of the enzyme and its clot specificity should be investigated to enhance therapeutic applicability.
The authors would like to acknowledge NTU, Faisalabad, Pakistan for providing facilities to carry out the zeta sizer and department of pharmacy, UAF, Pakistan for animal housing.
1. Zia MA. Streptokinase: An Efficient Enzyme in Cardiac Medicine. Protein & Peptide Letters. 2020; 27 (2). Available from: https://doi.org/10.2174/0929866526666191014150408
2. Khankari S, Badoei-Dalfard A, Karami Z. Cross-linked Enzyme Aggregates of Fibrinolytic Protease BC1 Immobilized on Magnetic Chitosan Nanoparticles (CLEAs-Fib-mChi): Synthesis, Purification, and Characterization. Applied Biochemistry and Biotechnology. 2021; 193 (6). Available from: https://doi.org/10.1007/s12010-021-03494-z
3. Vaishnavi B, Subathra Devi C. Potential application of immobilized streptokinase extracted from <i>Streptococcus equinus</i> VIT_VB2. Preparative Biochemistry & Biotechnology. 2017; 47 (10). Available from: https://doi.org/10.1080/10826068.2017.1373291
4. Kunamneni A, Abdelghani TTA, Ellaiah P. Streptokinase—the drug of choice for thrombolytic therapy. Journal of Thrombosis and Thrombolysis. 2007; 23 (1). Available from: https://doi.org/10.1007/s11239-006-9011-x
5. El-Dabaa E, Okasha H, Samir S, Nasr SM, El-Kalamawy HA, Saber M. Optimization of high expression and purification of recombinant streptokinase and in vitro evaluation of its thrombolytic activity. Arabian Journal of Chemistry. 2022; 15 (5). Available from: https://doi.org/10.1016/j.arabjc.2022.103799
6. Saxena K, Maturu VN, Maturu VN. A Comparative Study of the Safety and Efficacy of Intrapleural Fibrinolysis With Streptokinase and Urokinase in the Management of Loculated Pleural Effusions. Cureus. 2022; Available from: https://doi.org/10.7759/cureus.26271
7. Adzerikho IE, Vladimirskaya TE, Lutsik IL, Dubatouka KI, Agabekov VE, Branovitskay ES, <i>et al</i>. Fibrinspecific liposomes as a potential method of delivery of the thrombolytic preparation streptokinase. Journal of Thrombosis and Thrombolysis. 2022; 53 (2). Available from: https://doi.org/10.1007/s11239-021-02614-0
8. Hassan ME, Yang Q, Xiao Z. Impact of immobilization technology in industrial and pharmaceutical applications. 3 Biotech. 2019; 9 (12). Available from: https://doi.org/10.1007/s13205-019-1969-0
9. Srividya AR, Vishnuvarthan VJ. Immobilization Of Therapeutically Beneficial Enzymes. 2014.
10. Liu R, Luo C, Pang Z, Zhang J, Ruan S, Wu M, <i>et al</i>. Advances of nanoparticles as drug delivery systems for disease diagnosis and treatment. Chinese Chemical Letters. 2023; 34 (2). Available from: https://doi.org/10.1016/j.cclet.2022.05.032
11. Liu L, Li Y, AL-Huqail AA, Ali E, Alkhalifah T, Alturise F, <i>et al</i>. Green synthesis of Fe3O4 nanoparticles using Alliaceae waste (Allium sativum) for a sustainable landscape enhancement using support vector regression. Chemosphere. 2023; 334 Available from: https://doi.org/10.1016/j.chemosphere.2023.138638
12. Bilal M, Zhao Y, Rasheed T, Iqbal HM. Magnetic nanoparticles as versatile carriers for enzymes immobilization: A review. International Journal of Biological Macromolecules. 2018; 120 Available from: https://doi.org/10.1016/j.ijbiomac.2018.09.025
14. Masood U, Riaz R, Shah SU, Majeed AI, Abbas SR. Contrast enhanced sonothrombolysis using streptokinase loaded phase change nano-droplets for potential treatment of deep venous thrombosis. RSC Advances. 2022; 12 (41). Available from: https://doi.org/10.1039/d2ra04467f
15. Zhang B. Intravascular Sonothrombolysis with Magnetic Microbubbles and Nanodroplets. Dissertations-Theses. North Carolina State University. 2023.
16. Ghaffar A, Ahmed B, Munir B, Faisal R, Mahmood Z. Production and Characterization of Streptokinase Enzyme by Using Streptococcus mutans Strain in Liquid State Fermentation through Corn Steep Liquor (CSL) Substrate. Biochemistry & Physiology: Open Access. 2015; 04 (04). Available from: https://doi.org/10.4172/2168-9652.1000178
17. Naseem H, Pal ZAA, Salamat B, Jabbar A, Tarar MHA, Maratib M, <i>et al</i>. Effects of Different Parameters on the Production of Streptokinase Enzyme. Saudi Journal of Biomedical Research. 2021; 6 (5). Available from: https://doi.org/10.36348/sjbr.2021.v06i05.006
18. Ibrahim D. Statistical Study of Extract Keratin Protein from Waste Chicken Feather Based on Response Surface Methodology. Scientific Journal for the Faculty of Science-Sirte University. 2022; 2 (2). Available from: https://doi.org/10.37375/sjfssu.v2i2.76
19. Rogala A, Rechberger J, Vasold V, Samphao A, Kalcher K, Ortner A. Voltammetric lipase activity assay based on dilinolein and a modified carbon paste electrode. Analytical and Bioanalytical Chemistry. 2022; 414 (17). Available from: https://doi.org/10.1007/s00216-022-04135-y
20. Liudmyla K, Olena C, Nadiia S. Chemical properties of Helix aspersa mucus as a component of cosmetics and pharmaceutical products. Materials Today: Proceedings. 2022; 62 Available from: https://doi.org/10.1016/j.matpr.2022.02.217
13. da Silva MM, Neto JMWD, Regueira BVB, do Couto MTT, Sobral RVDS, Conniff AES, <i>et al</i>. Immobilization of fibrinolytic protease from Mucor subtilissimus UCP 1262 in magnetic nanoparticles. Protein Expression and Purification. 2022; 192 Available from: https://doi.org/10.1016/j.pep.2022.106044
21. Prabowo B, Khairunnisa T, Nandiyanto ABD. Economic perspective in the production of magnetite (Fe3O4) nanoparticles by co-precipitation method. World Chemical Engineering Journal. 2018;2(2):1-4.
22. Neto JMWD, de Albuquerque Wanderley MC, de Albuquerque Lima C, Porto ALF. Single step purification via magnetic nanoparticles of new broad pH active protease from Penicillium aurantiogriseum. Protein Expression and Purification. 2018; 147 Available from: https://doi.org/10.1016/j.pep.2018.01.016
23. Hachim LS, Aljoofy IK, Khalaf KJ, Abdul-Hussein SS. Immunomodulatory Activity of Staphylococcus aureus Purified Staphylokinase and Streptokinase in BALB/C mice. Research Journal of Pharmacy and Technology. 2023; 16 (2). Available from: https://doi.org/10.52711/0974-360x.2023.00143
24. Balaraman K, Prabakaran G. Production & purification of a fibrinolytic enzyme (thrombinase) from Bacillus sphaericus. Indian J Med Res. 2007;126(5):459-464. https://pubmed.ncbi.nlm.nih.gov/18160751/
25. Tanveer R, Gulzar H, Ahmad B, Akhtar M, Hussain I, Hanif Q, <i>et al</i>. Optimization of Metal Ions in Sugarcane Bagasse Fermenting Medium for the Production of Streptokinase by Streptococcus equisimilis. Science Letters. 2021; 9 (2). Available from: https://doi.org/10.47262/sl/9.2.132021003
26. Akbar G, Zia M, Jamil A, Joyia F. Overproduction Of Streptokinase from Streptococcus Mutans EBL-37-Uv90 By Performing Optimizational Studies. The Journal of Animal and Plant Sciences. 2021; 31 (6). Available from: https://doi.org/10.36899/japs.2021.6.0383
27. Ghosal T, Augustine N, Siddapur A, Babu V, Samuel MK, Chandrasekaran SD. Strain improvement, optimization and purification studies for enhanced production of streptokinase from Streptococcus uberis TNA-M1. Frontiers in Biology. 2017; 12 (5). Available from: https://doi.org/10.1007/s11515-017-1467-x
28. Tandon S, Sharma A, Singh S, Sharma S, Sarma SJ. Therapeutic enzymes: Discoveries, production and applications. Journal of Drug Delivery Science and Technology. 2021; 63 Available from: https://doi.org/10.1016/j.jddst.2021.102455
29. Narasimhan MK, Ethiraj S, Krishnamurthi T, Rajesh M. Purification, biochemical, and thermal properties of fibrinolytic enzyme secreted by<i>Bacillus cereus</i>SRM-001. Preparative Biochemistry & Biotechnology. 2018; 48 (1). Available from: https://doi.org/10.1080/10826068.2017.1387560
30. Brown JQ, Vishwanath K, Palmer GM, Ramanujam N. Advances in quantitative UV–visible spectroscopy for clinical and pre-clinical application in cancer. Current Opinion in Biotechnology. 2009; 20 (1). Available from: https://doi.org/10.1016/j.copbio.2009.02.004
© 2025 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.