

Journal of Pharmaceutical Research
DOI: 10.18579/jopcr/v22.2.23.5
Year: 2023, Volume: 22, Issue: 2, Pages: 55-62
Original Article
Senthil Kumar Raju ✉ 1 , Shridharshini Kumar 1 , Praveen Sekar 1 , Naveena Sundhararajan 1 , Yogadharshini Nagalingam 1
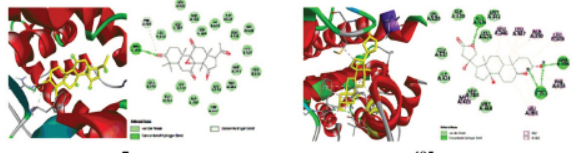
Breast cancer is one of the most common cancers in women all around the world and is a dominant cause of deaths occurring all around the globe. The available potent drugs for breast cancer show adverse effects and resistance and are found to be ineffective in patients. The high cost of currently available cancer therapy and certain limitations of current treatment make it necessary to search for novel, cost-effective and efficient methods of cancer treatment. Phytochemicals are directly involved in treatment or precursors to synthesize useful drugs. Therefore, in the current investigation, 500 terpenoid phytoconstituents and five proteins associated with breast cancer including EGFR, ERα, HER2, NF-κB and Topo IIa were selected from various databases. Selected compounds were screened for their molecular properties based on Lipinski's rule of five resulting in 235 compounds exclusion. Drug-likeness and PAINS alert properties were predicted using pkCSM and SwissADME web servers which led to the omission of 43 compounds. The remaining 222 compounds were screened to predict their ADMET properties and based on these results, 117 compounds were selected to predict the anti-breast cancer potential. Finally, 73 compounds, which showed anti-breast cancer activity prediction, were virtually screened and the top four best-scoring compounds were selected as lead-like molecules and docked with the five respective breast cancer targets. The results showed that the top four lead-like molecules exhibited greater binding affinity and lesser toxicity than the standard drugs namely 4–Hydroxytamoxifen, Daunorubicin, Erlotinib and Lapatinib.
Keywords: ADMET, Breast cancer, Chemotherapeutic agents, In silico analysis, Molecular docking, Terpenoids
© 2023 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.