

Journal of Pharmaceutical Research
Year: 2026, Volume: 25, Issue: 1, Pages: 53-56
Case Report
Bhaumikkumar A Patel1*, Anil P Singh2, Amita R Kubavat3, Hardip V Bakutra1
1Senior resident, Department of Pharmacology, PDU Medical College and Hospital, Rajkot, Gujarat
2Professor & Head of Department, Department of Pharmacology, PDU Medical College and Hospital, Rajkot, Gujarat
3Assistant professor, Department of Pharmacology, PDU Medical College and Hospital, Rajkot, Gujarat
*Corresponding Author
Email: [email protected]
Acute generalized exanthematous pustulosis (AGEP) is a rapid-onset pustular dermatosis characterized by widespread sterile pustules on erythematous skin, commonly triggered by medications. Etoricoxib, a selective COX-2 inhibitor widely used for inflammatory and pain conditions, is a rare but recognized cause of drug-induced AGEP. A female patient in her forties had prescribed oral etoricoxib 90 mg two times a day for the management of dental pain. After that patient started to develop multiple bilateral symmetrical pustules with erythematous base present over both bilateral upper limb and lower limb. Patient was treated with Inj. Hydrocortisone intravenous stat, Tab. Prednisolone 40 mg orally once a day, Tab. Azithromycin 500 mg once a day, Tab. Levocetirizine 5 mg orally twice a day and Tab. Pantoprazole 40 mg twice a day for 5 days. For pustules over skin patient was treated with fremycetin cream and betamethasone cream. Patient was recovered in seven days. The adverse drug reaction was assessed as certain by the WHO causality scale and probable by the Naranjo scale. Severity was categorized as moderate (Level 3) using the Modified Hartwig and Siegle scale, while preventability assessment indicated that the reaction was definitely preventable.
Keywords: Acute generalized Exanthematous Pustulosis, Etoricoxib
Acute generalized exanthematous pustulosis (AGEP) was initially described in 1968 by Baker and Ryan as a distinctive pustular drug reaction. Subsequently, in 1980, Beylot and colleagues introduced the term pustulose exanthémateuse aiguë généralisée (PEAG) in French literature, which later evolved into the widely accepted nomenclature “acute generalized exanthematous pustulosis.”AGEP is a rare dermatological condition with an estimated incidence of approximately 1–2 cases per million individuals per year. It is most commonly triggered by medications, although infections have also been reported as precipitating factors[1].
Clinically, AGEP presents as an acute onset of numerous small, sterile, non-follicular pustules arising on an erythematous background, often accompanied by fever and leukocytosis. Symptoms typically develop within 48 hours following exposure to the offending agent. In most patients, the disease follows a self-limiting course with spontaneous resolution after withdrawal of the causative drug. However, severe cases may involve mucosal surfaces and systemic organ dysfunction[2].
Etoricoxib is a selective cyclooxygenase-2 (COX-2) inhibitor widely prescribed for the management of inflammatory and painful conditions such as osteoarthritis, rheumatoid arthritis, acute gout, ankylosing spondylitis, dysmenorrhea, and postoperative dental pain. Its therapeutic advantage lies in reduced gastrointestinal toxicity and minimal interference with platelet function compared to non-selective nonsteroidal anti-inflammatory drugs. Common adverse effects associated with etoricoxib include abdominal discomfort, headache, dizziness, palpitations, constipation, and elevation of blood pressure[3, 4].
Additionally, etoricoxib has been linked to an increased risk of cardiovascular and cerebrovascular thrombotic events, heart failure, hypertension, and peripheral edema. Due to these safety concerns, the United States Food and Drug Administration (USFDA) advisory committee declined its approval in 2007, primarily because of its potential cardiovascular risks. In contrast, the drug has been approved in the United Kingdom for short-term use in acute pain and selected arthritic conditions[5].
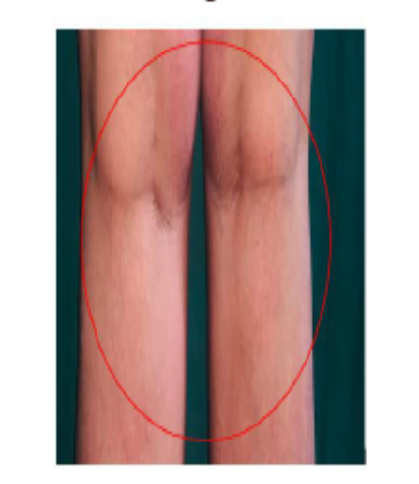
A female patient in her forties presented to the dermatology outpatient department with complaints of rapidly progressing erythematous skin lesions. The initial erythema appeared on the right lower limb and within approximately three hours extended to involve the left lower limb. By the following day, multiple small, symmetrical pustules developed over the erythematous areas on both legs.
After the appearance of pustules, the patient experienced intense pruritus followed by a burning sensation over the affected regions. On further history taking, it was revealed that she had been prescribed oral etoricoxib 90 mg two time a day by a private practitioner for dental pain shortly before the onset of symptoms.
Based on the characteristic clinical features, EuroSCAR criteria and temporal association with drug exposure, the reaction was diagnosed as AGEP. The patient had no significant comorbid conditions; however, a past history of a similar cutaneous reaction following etoricoxib intake was noted.
Etoricoxib was immediately discontinued. The patient was managed on an outpatient basis with intravenous hydrocortisone 100 mg administered stat, followed by oral tablet prednisolone 40 mg once a day. Additional supportive therapy included tablet azithromycin 500 mg once a day, tablet levocetirizine 5 mg once a day, and tablet pantoprazole 40 mg two times a day for five days. Topical treatment consisting of Fremycetin cream and betamethasone cream was applied to the pustular lesions.
The patient showed gradual clinical improvement, with complete resolution of lesions within seven days of treatment initiation.
The patient presented to the OPD three days following initiation of treatment for AGEP, demonstrating signs indicative of the recovery phase. By the seventh day post-treatment, the patient had fully recovered from the condition.
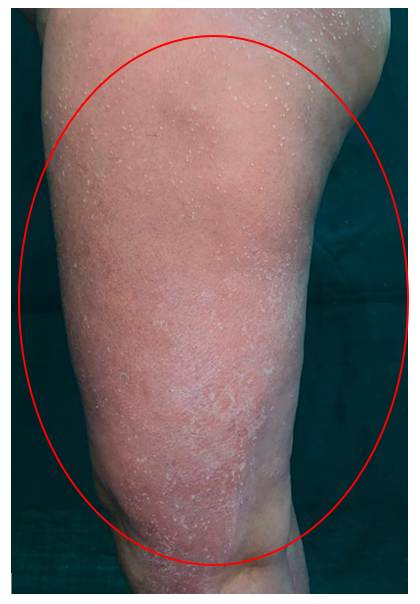
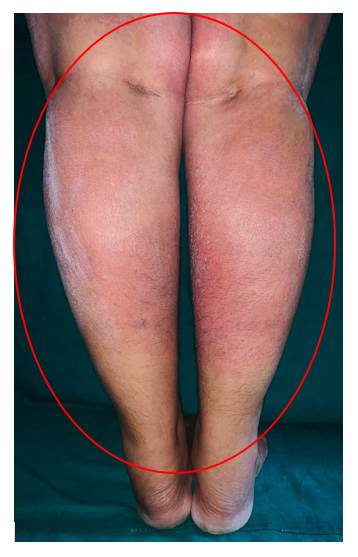
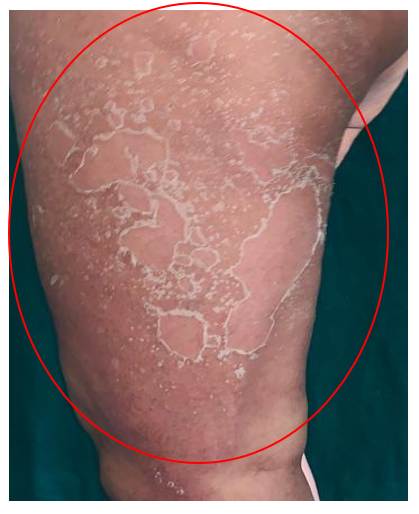
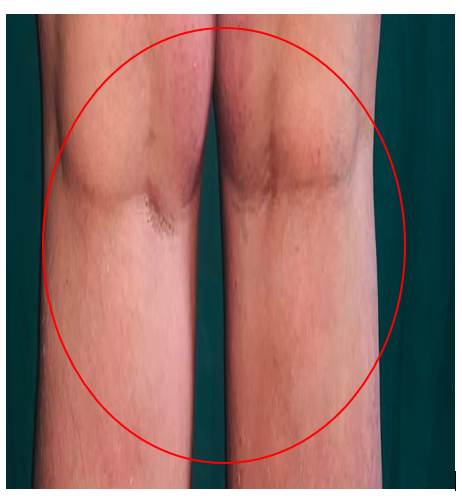
According to data available from VigiAccess up to May 2024, a total of 25,745 adverse drug reaction reports related to etoricoxib have been documented, of which approximately 32% involved cutaneous manifestations. This highlights the significant association between etoricoxib use and skin-related adverse events. Furthermore, the EuroSCAR study reported 97 confirmed cases of AGEP from multiple European countries, with a higher predominance among female patients and a mean age of 56 years[2].
Comparable demographic patterns have been observed in studies conducted in Israel and Taiwan, where AGEP was more frequently reported in females, typically presenting at a younger mean age of around 40 years. Although AGEP is more common in adults, pediatric cases have also been documented, including a notable case series of 20 children reported from China, indicating that the condition can affect all age groups[2].
The onset of AGEP is generally rapid following exposure to the causative drug, most commonly occurring within 1 to 5 days. However, the latency period may vary depending on the pharmacological class of the triggering agent. For instance, antibiotics such as sulfonamides tend to induce symptoms within 24 hours, whereas other medications may exhibit a delayed onset of up to 11 days. The patient also received concomitant medications, including hydrocortisone, prednisolone, levocetirizine, pantoprazole, and azithromycin; however, these were initiated after the onset of skin lesions and are less commonly associated with AGEP, making them unlikely confounding factors in this scenario. Some studies have also suggested seasonal variability in AGEP occurrence, with higher clustering of cases during summer months, particularly in Israel. This seasonal trend corresponds with the overall higher prevalence of drug-induced cutaneous reactions among female patients[2].
AGEP is classified as a type IV hypersensitivity reaction and is regarded as a distinct clinical and histopathological entity. The condition is characterized by an abrupt onset of edematous erythema followed by the rapid appearance of numerous small, superficial, non-follicular sterile pustules, typically measuring less than 5 mm in diameter. These cutaneous manifestations generally resolve spontaneously within two weeks, frequently accompanied by post-inflammatory desquamation[6, 7].
Histopathological examination commonly reveals spongiform subcorneal and/or intraepidermal pustules, marked edema of the papillary dermis, and a mixed perivascular inflammatory infiltrate predominantly composed of neutrophils, with occasional eosinophils exhibiting epidermal exocytosis. Application of the EuroSCAR scoring system categorized the case as “probable AGEP,” reinforcing the diagnostic validity. These findings support the diagnosis and help differentiate AGEP from other pustular dermatoses[8].
Although AGEP is most often drug-induced, various infectious agents—including viral pathogens, bacterial organisms such as Streptococcus, and parasitic infections like Echinococcus—as well as exposure to mercury have also been implicated as potential triggers. However, pharmacological agents remain the predominant cause, accounting for approximately 90% of reported cases. Frequently associated medications include antimicrobials, particularly aminopenicillins and macrolides, antifungal agents such as terbinafine, itraconazole, and nystatin, tetracyclines, allopurinol, calcium channel blockers, and paracetamol[9].
From an immunological perspective, AGEP involves drug-specific T lymphocytes that produce elevated levels of chemokine (C-X-C motif) ligand 8, also known as interleukin-8, which plays a crucial role in neutrophil recruitment and subsequent pustule formation. The accumulation of neutrophils within the epidermis is a hallmark feature of the disease. It has been suggested that activated CD4+ T cells, along with a subset of CD8+ T cells, act as primary mediators in the early phase of the reaction and contribute to vesiculopustular lesion development[6].
Additionally, Roujeau and colleagues reported a higher-than-expected occurrence of AGEP in individuals with a personal or family history of psoriasis vulgaris, suggesting a possible genetic or immunological predisposition contributing to disease susceptibility[1].
The adverse drug reaction is deemed certain according to the WHO causality assessment scale, as the patient had previously experienced similar symptoms from etoricoxib[10]. According to the Naranjo probability assessment scale, the reaction is considered Probable[11]. The Modified Hartwig and Siegle Severity scale rates the reaction as Moderate (Level-3)[12]. Furthermore, the Modified Schumock & Thornton Preventability score indicates that the reaction could have been definitely Prevented[13].
It highlights the need for clinicians to maintain a high index of suspicion when evaluating acute pustular eruptions, particularly in patients with a history of exposure to the suspected drug. Importantly, this case underscores the necessity of prompt drug discontinuation and strict avoidance of re-exposure in sensitized individuals to prevent recurrence. Continued reporting of such rare adverse drug reactions remains essential to strengthen pharmacovigilance data and support safer clinical practice.
The authors would like to express their sincere gratitude to the Department of Dermatology, PDU Medical College and Civil Hospital, Rajkot, for their valuable support and cooperation in the diagnosis and management of this case. We are especially thankful to the former Head of the Department, Dr. Neela Bhuptani, for her guidance, encouragement, and academic support.
We also extend our appreciation to all colleagues and staff members who provided assistance and support during the preparation of this case report.
Finally, the author expresses heartfelt gratitude to his parents and wife for their constant encouragement, understanding, and unwavering support throughout this work.
AGEP- Acute generalize exanthematous pustulosis; PEAG- pustulose exanthémateuse aiguë généralisée; COX-2- Cyclooxygenase-2; USFDA- United states food and drug administration; OPD- Outpatients department
1. Acute Generalized Exanthematous Pustulosis. Archives of Dermatology. 1991; 127 (9). Available from: https://doi.org/10.1001/archderm.1991.01680080069004
2. Controversies in the management of cutaneous adverse drug reactions. Indian Journal of Dermatology. 2018; 63 (2). Available from: https://doi.org/10.4103/ijd.ijd_585_17
3. Etoricoxib-induced Acute Generalized Exanthematous Pustulosis. Acta Dermato-Venereologica. 2008; 88 (2). Available from: https://doi.org/10.2340/00015555-0381
4. Pakdeethai J, Ho SA, Aw D, Bing Tan K. <I>Therapeutic hotline Acute generalized exanthematous pustulosis-like, folliculitic drug reaction pattern caused by celecoxib</I>. 2012.
5. The cardiovascular risks of etoricoxib (<i>Arcoxia</i>). Annals of Saudi Medicine. 2008; 28 (2). Available from: https://doi.org/10.4103/0256-4947.51740
6. Valdecoxib-associated acute generalized exanthematous pustulosis. Burns. 2005; 31 (3). Available from: https://doi.org/10.1016/j.burns.2004.10.017
7. Acute generalized exanthematous pustulosis: report of 12 cases and literature review. International Journal of Dermatology. 2009; 48 (3). Available from: https://doi.org/10.1111/j.1365-4632.2009.03908.x
8. Acute Generalized Exanthematous Pustulosis: Clinical Features, Differential Diagnosis, and Management. American Journal of Clinical Dermatology. 2023; 24 (4). Available from: https://doi.org/10.1007/s40257-023-00779-3
9. Drug Triggers and Clinic of Acute Generalized Exanthematous Pustulosis (AGEP): A Literature Case Series of 297 Patients. Journal of Clinical Medicine. 2022; 11 (2). Available from: https://doi.org/10.3390/jcm11020397
10. World Health Organization. <I>The use of the WHO-UMC system for standardised case causality assessment</I>. Geneva: World Health Organization.
11. <I>Canadian Medical Association Journal. Case 3. Ottawa: CMAJ</I>; 2022 Apr 20. Available from: https://www.cmaj.ca/content/cmaj/suppl/2022/04/20/194.16.E581.DC3/211687-case-3-at.pdf
12. Preventability and severity assessment in reporting adverse drug reactions. American Journal of Health-System Pharmacy. 1992; 49 (9). Available from: https://doi.org/10.1093/ajhp/49.9.2229
13. Schumock GT, Thornton JP. Focusing on the preventability of adverse drug reactions. <I>Hospital Pharmacy</I>. 1992;27(6):538. https://pubmed.ncbi.nlm.nih.gov/10118597/
© 2026 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.