

Journal of Pharmaceutical Research
Year: 2026, Volume: 25, Issue: 1, Pages: 16-19
Review Article
Sagarika Basavaraj1*, Aparna Chincholkar2
1Junior Resident, Department of Pharmacology, MAEER'S MIT Pune's MIMER Medical College and Dr BSTR Hospital, Talegaon Dhabade, Pune- 410507, Maharashtra, India
2Professor, Department of Pharmacology, MAEER'S MIT Pune's MIMER Medical College and Dr BSTR Hospital, Talegaon Dhabade, Pune- 410507, Maharashtra, India
*Corresponding Author
Sagarika Basavaraj
Email: [email protected]
Obesity is a chronic, relapsing disease associated with significant cardiometabolic, mechanical, and psychological consequences. Despite lifestyle interventions forming the cornerstone of therapy, long-term weight loss is difficult to sustain for many individuals, leading to growing interest in pharmacological options. Semaglutide, a long-acting glucagon-like peptide-1 receptor agonist, has emerged as an important therapeutic advancement due to its substantial weight-reduction effects and metabolic benefits. The objective of the study is to describe the clinical, pharmacological, and therapeutic profile of semaglutide in the context of obesity management, including mechanism of action, pharmacokinetic characteristics, efficacy, safety considerations, dosing strategies, and future directions. Relevant literature from clinical trials, mechanistic studies, regulatory reports, and real-world analyses was reviewed to summarize the therapeutic role of semaglutide in obesity. Particular attention was given to major clinical trial programs evaluating weight outcomes, metabolic parameters, and safety events. Semaglutide promotes weight loss through central and peripheral mechanisms that reduce appetite, enhance satiety, delay gastric emptying, and improve glucose homeostasis. Molecular modifications prolong its half-life, enabling convenient once-weekly dosing and consistent metabolic effects. Clinical evidence shows notable reductions in body weight, improvements in glycemic control, and favorable cardiometabolic changes in diverse patient groups. The most common adverse effects are gastrointestinal and tend to lessen with gradual dose escalation. Concerns such as pancreatitis, gallbladder disease, and thyroid-related risks are uncommon but require clinical awareness. High-dose oral formulations, extended indications, and combination therapies are under ongoing investigation and may further broaden clinical utility. Semaglutide represents a significant evolution in the medical treatment of obesity, offering durable weight loss and meaningful metabolic benefits. While cost, accessibility, and long-term safety considerations persist, current evidence supports its role as a cornerstone.
Keywords: Semaglutide, Obesity, Pharmacotherapy, GLP-1 receptor agonist, Weight management, Metabolism
Obesity is defined as an abnormal or excessive accumulation of body fat that impairs health, with a body mass index (BMI) ≥30 kg/m² widely accepted as the diagnostic threshold[1]. The severity of obesity is categorized as Class I (BMI 30–34.9 kg/m²), Class II (35–39.9 kg/m²), and Class III (≥40 kg/m²), with higher classes conferring progressively greater morbidity risk[2]. The global prevalence of obesity has risen sharply, with recent pooled analyses showing widespread increases across 200+ countries over the past three decades[3]. In India, the ICMR–INDIAB 2023 study reported a combined overweight-and-obesity prevalence of 28.6%, highlighting the growing burden in both urban and rural populations[3]. Urban tertiary hospital studies indicate even higher rates, frequently ranging from 31% to 45%, reflecting lifestyle transitions, socioeconomic factors and reduced physical activity in metropolitan areas.³ Obesity contributes to a spectrum of complications including type 2 diabetes mellitus, hypertension, dyslipidemia, coronary artery disease, osteoarthritis, obstructive sleep apnea and non-alcoholic fatty liver disease, thereby increasing all-cause mortality and posing significant public health challenges[4]. Metabolic syndrome, a closely related condition characterized by central obesity, insulin resistance, dyslipidemia and hypertension, further amplifies cardiometabolic risk[5].
The limited sustainability of lifestyle interventions alone and the chronic, relapsing nature of obesity have intensified the need for effective pharmacotherapies[6]. Semaglutide, a long-acting glucagon-like peptide-1 receptor agonist (GLP-1 RA), represents a major advancement in medical obesity management due to its substantial and durable weight-loss efficacy demonstrated across diverse populations[6]. The drug acts on key central and peripheral pathways regulating appetite, satiety and glucose homeostasis, offering meaningful improvements in metabolic parameters and obesity-related comorbidities[6]. With a rapidly expanding evidence base and regulatory approval for chronic weight management, semaglutide has become a cornerstone of modern pharmacological treatment strategies for obesity[6].
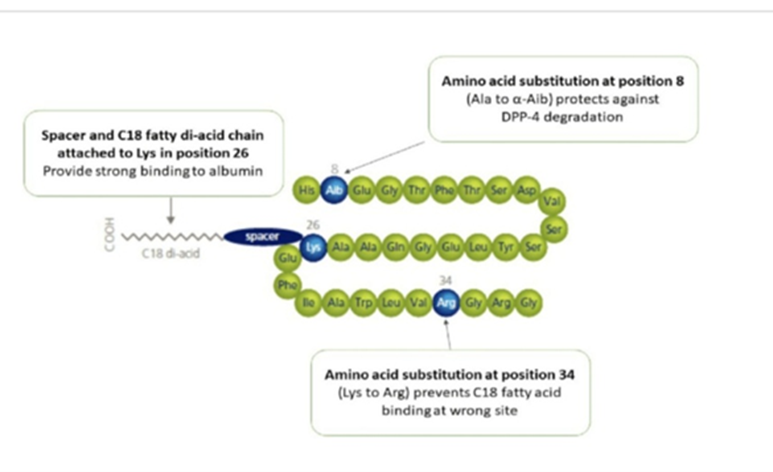
Semaglutide is a structurally modified analogue of human GLP-1, incorporating specific amino acid substitutions and acylation with a C18 fatty-diacid side chain to enhance albumin binding and resist degradation by dipeptidyl peptidase-4 (DPP-4)[7]. These modifications prolong its half-life, enabling once-weekly dosing and sustained activation of GLP-1 receptors, thereby improving therapeutic adherence and metabolic efficacy[7].
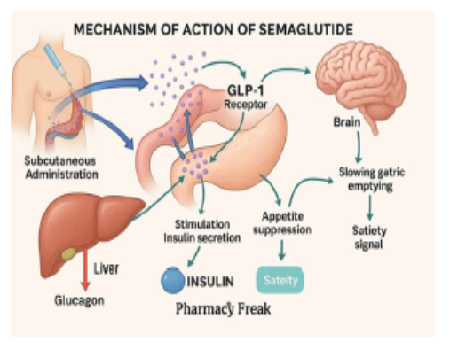
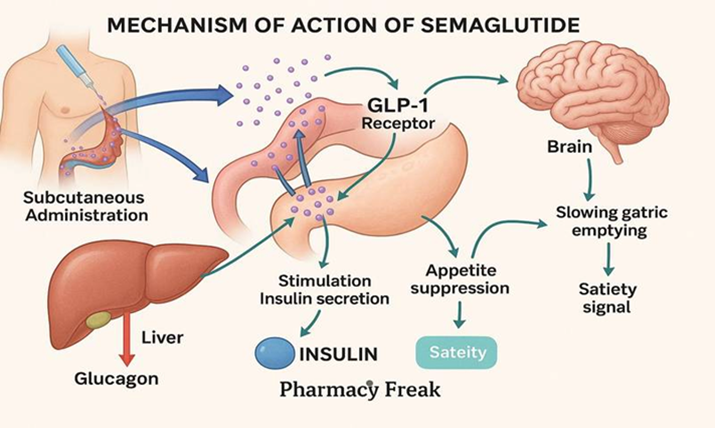
Semaglutide exerts its effects by binding to GLP-1 receptors in the hypothalamus, promoting satiety, reducing appetite and lowering caloric intake[8]. It delays gastric emptying, enhances glucose-dependent insulin secretion, suppresses glucagon release and improves pancreatic β-cell responsiveness[8]. Additionally, its central effects extend to reward pathways, reducing cravings and hedonic eating, which contributes significantly to weight loss in individuals with obesity[8].
Semaglutide exhibits high bioavailability with subcutaneous administration, achieving slow and sustained absorption due to strong albumin binding, which prolongs its half-life to approximately one week[9]. Its pharmacokinetics remain consistent across age groups and in mild-to-moderate renal or hepatic impairment, allowing predictable drug exposure without major dose adjustments[9]. Oral semaglutide utilizes absorption enhancer Salcaprozate sodium (SNAC) to facilitate gastric mucosal uptake, although with lower bioavailability than injectable preparations[9].
Following systemic absorption, semaglutide undergoes proteolytic degradation and β-oxidation rather than cytochrome P450 metabolism, minimizing the risk of drug–drug interactions[9]. Excretion occurs primarily through the urine and feces as inactive metabolites, and steady-state concentrations are achieved after four to five weeks of weekly dosing, consistent with its extended half-life[10].
Gastrointestinal adverse effects such as nausea, vomiting, diarrhea and constipation are the most common and arise from delayed gastric emptying and increased central satiety signaling[12]. Alopecia has been reported infrequently and is thought to result from rapid weight loss–induced telogen effluvium rather than a direct drug effect[12]. Ocular adverse effects, including transient blurred vision, may occur due to rapid glycemic improvements altering lens hydration dynamics[12]. Pancreatitis is a rare but serious concern, potentially linked to GLP-1–related effects on pancreatic exocrine cells and gallstone formation from weight loss[12]. Thyroid-related risks, including C-cell hyperplasia observed in rodents, underline theoretical concerns, although human evidence remains limited[12].
| Trial Name | Population | Dose | Outcome | Inference |
|---|---|---|---|---|
| STEP 1, 2, 3, 4[11] | Adults with overweight / obesity |
2.4 mg weekly |
~10–15% weight loss |
Robust and sustained weight reduction across cohorts. |
| SUSTAIN Trials[11] | Type 2 diabetes | 0.5–1 mg weekly |
Improved glycemic control + modest weight loss |
Effective for diabetes with metabolic benefits. |
| STEP 2 Trial[14] | T2DM + obesity | 2.4 mg weekly |
Greater weight loss vs placebo and 1 mg |
Confirms efficacy of higher-dose semaglutide in diabetes-associated obesity. |
Semaglutide is contraindicated in individuals with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 due to preclinical findings of C-cell tumors in rodents[13]. It should be avoided in patients with severe gastrointestinal disorders such as gastroparesis, as delayed gastric emptying may worsen symptoms[13]. Hypersensitivity to the formulation constitutes another contraindication[13]. Use during pregnancy is discouraged because intentional weight loss is contraindicated and human fetal safety data remain insufficient[13].
For obesity management, semaglutide is initiated at 0.25 mg weekly and titrated gradually to the target dose of 2.4 mg weekly to minimize gastrointestinal intolerance[14]. Oral semaglutide follows a stepwise increase to 14 mg daily for glycemic control, while higher-dose oral formulations for obesity are under active evaluation[14].
Cost-effectiveness analyses indicate that semaglutide 2.4 mg is economically viable in high-income countries due to reductions in diabetes progression, cardiovascular events and healthcare utilization[15]. However, its high retail cost remains a barrier in resource-limited settings, highlighting the need for pricing negotiations and broader insurance coverage to improve accessibility[15].
Future directions include expanded indications for obesity with comorbidities like metabolic liver disease, heart failure and addiction disorders[16]. Oral semaglutide (Rybelsus) formulations currently available include 3 mg, 7 mg and 14 mg tablets, with higher-dose tablets 25 mg and 50 mg are under investigation in OASIS trials for improved weight-loss efficacy[16]. Pen-injector (Ozempic) semaglutide is available as 0.25 mg, 0.5 mg, 1 mg and 2.4 mg pre-filled pens designed for once-weekly dosing[16]. Ongoing innovations aim to improve delivery systems, enhance potency and develop combination therapies to further optimize outcomes[16]. Possible fixed drug combinations are under investigation:
Semaglutide + Amylin analog (enhanced appetite suppression)
Semaglutide + GIP/GLP-1 dual agonist (complementary metabolic effects)
Semaglutide + SGLT2 inhibitor (cardiometabolic additive effect)
1. Rubino F, Cummings DE, Eckel RH, Ryan DH, Mechanick JI, Dixon JB, <I>et al</I>. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinology. 2025; 13 (3). Available from: https://doi.org/10.1016/s2213-8587(24)00316-4
2. Zahid S, Peng AW, Razavi AC, Moore JX, Shimbo D, Brown TM, <I>et al</I>. Center Stage: Putting Obesity Staging Systems Into the Spotlight. Preventing Chronic Disease. 2025; 22 Available from: https://doi.org/10.5888/pcd22.250222
3. NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies. Lancet. 2024; 403 (10431). Available from: https://doi.org/10.1016/S0140-6736(23)02750-2
4. Blüher M. An overview of obesity‐related complications: The epidemiological evidence linking body weight and other markers of obesity to adverse health outcomes. Diabetes, Obesity and Metabolism. 2025; 27 (S2). Available from: https://doi.org/10.1111/dom.16263
5. Peterseim CM, Koltookian K, Vrettos B. Metabolic Syndrome: An Updated Review on Diagnosis and Treatment for Primary Care Clinicians. Journal of Primary Care & Community Health. 2024; 15 Available from: https://doi.org/10.1177/21501319241309168
6. Gundapaneni SRC, Burri RG, Kaku R, Mamytova A, Kondadasula P, Tagaev T. Semaglutide in Obesity and Type 2 Diabetes Management: A Systematic Review of Clinical Outcomes. Cureus. 2025; 17 (2). Available from: https://doi.org/10.7759/cureus.78555
7. Mahapatra S, Damodaran A. Semaglutide: a GLP-1 receptor agonist with cardiovascular benefits in type 2 diabetes management. <I>Journal of Diabetes and its Complications</I>. 2022;36(4):108-115.
8. Papakonstantinou E, Partsinevelos G, Voulgaridou A, Trovas G, Lambrinoudaki I, Alexandrou A, <I>et al</I>. Mechanistic insights of semaglutide: from glucose control to pleiotropic metabolic effects. <I>Metabolites</I>. 2024;14(8):1-15.
9. Yang XD, Yang YY. Clinical Pharmacokinetics of Semaglutide: A Systematic Review. Drug Design, Development and Therapy. 2024; 18 Available from: https://doi.org/10.2147/dddt.s470826
10. Xie P, Abildlund MT, Bækdal TA, He X, Lyauk YK, Patted URH, <I>et al</I>. A phase 1, randomized, double-blind, placebo-controlled trial investigating the pharmacokinetics, pharmacodynamics, safety and tolerability of oral semaglutide in healthy Chinese subjects. Diabetes, Obesity and Metabolism. 2024; 26 (8). Available from: https://doi.org/10.1111/dom.15624
11. Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, <I>et al</I>. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine. 2021; 384 (11). Available from: https://doi.org/10.1056/nejmoa2032183
12. Sillassen CDB, Bjerregaard CD, Bjerre J, Wetterslev J, Jakobsen JC. The adverse effects associated with semaglutide use in patients at increased risk of cardiovascular events: a systematic review with meta-analysis and Trial Sequential Analysis. BMC Medicine. 2025; 23 (1). Available from: https://doi.org/10.1186/s12916-025-04486-0
13. Pillarisetti L, Agrawal DK. Semaglutide: benefits, risks and clinical considerations. <I>Archives of Internal Medicine Research</I>. 2025;8(1):1-13.
14. Davies M, Færch L, Jeppesen OK, Pakseresht A, Pedersen SD, Perreault L, <I>et al</I>. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. The Lancet. 2021; 397 (10278). Available from: https://doi.org/10.1016/s0140-6736(21)00213-0
15. Kim N, Wang J, Burudpakdee C, Song Y, Ramasamy A, Xie Y, <I>et al</I>. Cost-effectiveness analysis of semaglutide 2.4 mg for the treatment of adult patients with overweight and obesity in the United States. Journal of Managed Care & Specialty Pharmacy. 2022; 28 (7). Available from: https://doi.org/10.18553/jmcp.2022.28.7.740
16. Weiskirchen R. Semaglutide from bench to bedside: therapeutic evolution and future directions. <I>Medicina (Kaunas)</I>. 2025;61(4):265.
© 2026 Published by Krupanidhi College of Pharmacy. This is an open-access article under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/)
Subscribe now for latest articles and news.